Advertisements
Advertisements
प्रश्न
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
Advertisements
उत्तर
i. The ether (A) with molecular formula C5H12O is
\[\begin{array}{cc}
\ce{H3C - CH2 - O - HC -CH3}\\
\phantom{..............}|\\
\phantom{................}\ce{CH3}\\
\end{array}\]
ii. Reacts with hot HI to produce two alkyl halides as follows:

iii. Oxidation of (B) gives acid
\[\ce{CH3 - CH2 - OH ->[{[O]}] \underset{(D)}{CH3COOH}}\]
iv. Oxidation of (C) gives ketone
\[\begin{array}{cc}
\ce{CH3 - CH - OH ->[{[O]}] CH3 - C = O}\\
\phantom{...}|\phantom{....................}|\\
\phantom{....}\ce{CH3}\phantom{...........}\ce{(E)}\phantom{...}\ce{CH3}\\
\end{array}\]
Hence, structural formulae of compounds (A) to (E) are
(A) (2-Ethoxypropane)
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH3}\\
\phantom{.....................}|\phantom{...........}\\ \phantom{.......................}\ce{CH3\phantom{...........}}\\
\end{array}\]
(B) (Ethanol)
CH3CH2–OH
(c) (Propan-2-ol)
\[\begin{array}{cc}
\ce{CH3 - CH - CH3}\\
\phantom{..}|\\
\phantom{....}\ce{OH}\\
\end{array}\]
(D) (Ethanoic acid)
CH3COOH
(E) (Propanone)
\[\begin{array}{cc}
\ce{CH3 - C = O}\\
\phantom{...}|\phantom{.}\\
\phantom{....}\ce{CH3}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write two points of difference between the properties of phenol and ethyl alcohol.
Reaction between Grignard reagent and aldehyde other than formaldehyde leads to formation of _______________
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
Arrange the following in decreasing order of acid strength.
CH3OH, CH3–CH2–OH, CH3–CH(OH)–CH3, (CH3)3–C–OH
With the help of chemical equations show what happens when cumene hydroperoxide is treated with dil. acid.
How will you bring about the following conversions?
2-methyl propan-2-ol to 2-methylpropene
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
Which of the following reagents are required in the conversion of phenol to salicylic acid?
Which of the following compounds is obtained, when phenol react with bromine water?
α-butylene when subjected to hydroboration oxidation reaction, yields ______.
Bromination of phenol, will NOT give:
Which of following elements does not react with hot concentrated sulphuric acid?
Identify 'A' and 'B' in the following series of reactions.
Which of the following is INCORRECT regarding phenol?
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.

Identify reagents X, Y and Z.
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
Which of the following alcohols is least soluble in water?
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Identify product B in the following conversion?
\[\ce{Phenol ->[NaOH] A ->[CH3I] B}\]
A reaction of phenol with chloroform in presence of sodium hydroxide to form salicylaldehyde is known as ____________.

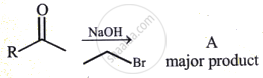
Product (B) in this reaction is:
Amongst the following alcohols which would react fastest with cone. HCl and ZnCl2?
What is the action of nitrous acid on aniline?
Write the chemical reaction when hot copper is treated with Vapours of 2° (secondary) alcohol.
Write chemical reaction when hot copper is treated with Vapours of 3° (tertiary) alcohol.
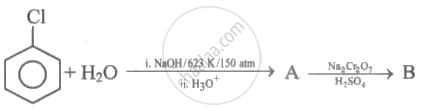
The structure of A in the given reaction is: