Advertisements
Advertisements
प्रश्न
What is the action of conc. H2SO4 on carbolic acid at 373 K.
Advertisements
उत्तर
At 373 K, carbolic acid (phenol) reacts with concentrated sulphuric acid to form p-phenolsulfonic acid.

APPEARS IN
संबंधित प्रश्न
Write two points of difference between the properties of phenol and ethyl alcohol.
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
With the help of chemical equations show what happens when cumene hydroperoxide is treated with dil. acid.
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
Write the reactions for the preparation of carbolic acid from aniline.
Which of the following compounds does not react with bromine in alkaline medium?
Propane when treated with cold cone. H2SO4 forms a compound which on heating with water gives ______.
Cumene is used in the commercial method for the manufacture of ____________.
The product 'C' in the following reaction is:
\[\ce{CH3CH2Br ->[alc. KCN] {'A'} ->[H3O^+][\Delta] {'B'} ->[i. LiAlH4][ii. H3O^+] {'C'}}\]
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
Which of the following alcohols has tertiary allylic carbon?
Which of the following reagents is used to convert phenol to benzene?
What is INCORRECT about the product written in the below given reaction?
\[\ce{R - CH2 - OH ->[PCC] R - COOH}\]
Which among the following is allylic secondary alcohol?
A reaction of phenol with chloroform in presence of sodium hydroxide to form salicylaldehyde is known as ____________.
Identify the compound having highest boiling point from the following?
The compound which reacts fastest with Lucas reagent at room temperature is ______.
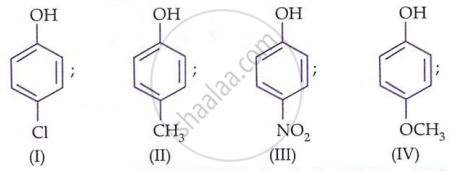
Which among the following phenolic compound is most acidic in nature?
Name the catalyst used in commercial method of preparation of phenol.

Product (B) in this reaction is:
Which among the following is not the method of preparation of phenol?
Write the chemical reaction when hot copper is treated with Vapours of 2° (secondary) alcohol.
Arrange the following compounds in order of decreasing acidity:
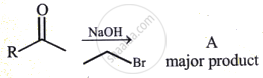
The structure of A in the given reaction is:
Which of the following is primary benzylic alcohol?
