Advertisements
Advertisements
प्रश्न
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
Advertisements
उत्तर
Primary, secondary and tertiary alcohols can be distinguished from each other in the laboratory using Lucas reagent (conc. HCl and ZnCl2).
- An unknown alcohol is treated with Lucas reagent.
The reaction involved is: \[\ce{R - OH ->[HCl][ZnCl2] R - Cl}\] - Depending on the observation an unknown alcohol can be identified as primary, secondary or tertiary.
Test Observation Inference Unknown Alcohol + Lucas reagent Reagent turns turbid on heating. Primary alcohol is present. Unknown Alcohol + Lucas reagent Reagent turns turbid slowly, without heating. Secondary alcohol is present. Unknown Alcohol + Lucas reagent Reagent turns turbid immediately, without heating. Tertiary alcohol is present. - The reaction between isopropyl alcohol and Lucas reagent can be given as:
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 + HCl + ZnCl2 -> CH3 - CH - CH3}\\
\phantom{..}|\phantom{......................................}|\phantom{..}\\
\phantom{.....}\ce{\underset{\text{Isopropyl alcohol (2° alcohol)}}{OH} \phantom{....................}\underset{\text{Isopropyl chloride}}{Cl}}\phantom{........}
\end{array}\]
The reagent will turns turbid slowly, without heating.
APPEARS IN
संबंधित प्रश्न
Write two points of difference between the properties of phenol and ethyl alcohol.
Answer in brief.
Give the reagents and conditions necessary to prepare phenol from Benzene sulfonic acid.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
With the help of chemical equations show what happens when cumene hydroperoxide is treated with dil. acid.
Draw intramolecular hydrogen bonding structure in o-nitrophenol.
Write chemical equation of acetyl chloride with phenol
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
Write the reactions for the preparation of carbolic acid from aniline.
What is the action of conc. H2SO4 on carbolic acid at 373 K.
Write chemical reactions to convert phenol into salicyaldehyde. Write the name of the reaction. What happens when if CCl4 is used instead of CHCl3 in the above reaction.
Number of oxygen atoms present in salicylaldehyde are ______.
Which alcohol is difficult to oxidise?
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
Which of the following reagents are required in the conversion of phenol to salicylic acid?
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
Cumene is used in the commercial method for the manufacture of ____________.
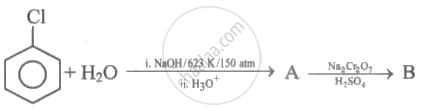
Identify 'A' and 'B' in the following series of reactions.
Which isomer of C4H10O is optically active?
Which of the following alcohols is least soluble in water?
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
The acid, which contains both -OH and -COOR groups is ______.
The order of reactivity of hydrogen halides with ether is as follows.
Write the product when 1°, 2° and 3° alcohol vapours are passed over hot copper.
Convert the following :
cumene to phenol.
What is the name of isopropyl alcohol according to the carbinol system?
Which of the following is trihydric phenol?
