Advertisements
Advertisements
Question
An unknown alcohol is treated with Lucas reagent. Explain how you will determine whether the alcohol is primary, secondary or tertiary. Indicate by chemical equation the reaction between isopropyl alcohol and Lucas reagent.
Advertisements
Solution
Primary, secondary and tertiary alcohols can be distinguished from each other in the laboratory using Lucas reagent (conc. HCl and ZnCl2).
- An unknown alcohol is treated with Lucas reagent.
The reaction involved is: \[\ce{R - OH ->[HCl][ZnCl2] R - Cl}\] - Depending on the observation an unknown alcohol can be identified as primary, secondary or tertiary.
Test Observation Inference Unknown Alcohol + Lucas reagent Reagent turns turbid on heating. Primary alcohol is present. Unknown Alcohol + Lucas reagent Reagent turns turbid slowly, without heating. Secondary alcohol is present. Unknown Alcohol + Lucas reagent Reagent turns turbid immediately, without heating. Tertiary alcohol is present. - The reaction between isopropyl alcohol and Lucas reagent can be given as:
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 + HCl + ZnCl2 -> CH3 - CH - CH3}\\
\phantom{..}|\phantom{......................................}|\phantom{..}\\
\phantom{.....}\ce{\underset{\text{Isopropyl alcohol (2° alcohol)}}{OH} \phantom{....................}\underset{\text{Isopropyl chloride}}{Cl}}\phantom{........}
\end{array}\]
The reagent will turns turbid slowly, without heating.
APPEARS IN
RELATED QUESTIONS
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Answer in brief.
Explain why p-nitrophenol is a stronger acid than phenol.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
When vapours of tert.butyl alcohol are passed over hot copper, it gives _____________
Write the reaction to get aspirin from salicylic acid.
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
Which alcohol is difficult to oxidise?
In the Lucas test for alcohols, the appearance of turbidity is due to the formation of ____________.
Which of the following compounds is obtained, when phenol react with bromine water?
The CORRECT decreasing order of boiling points for isomeric primary (1°), secondary (2°) and tertiary (3°) alcohols is ____________.
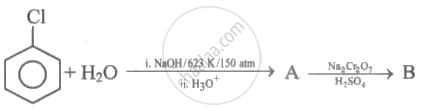
Identify 'A' and 'B' in the following series of reactions.
The product 'C' in the following reaction is:
\[\ce{CH3CH2Br ->[alc. KCN] {'A'} ->[H3O^+][\Delta] {'B'} ->[i. LiAlH4][ii. H3O^+] {'C'}}\]
____________ will NOT undergo acetylation.

Identify reagents X, Y and Z.
\[\ce{Isopropyl alcohol + acidic K2Cr2O7 -> X}\]
Identify product 'X' in the above reactions.
(I)

(II) \[\ce{P + Zn ->[\Delta] Q}\]
(III) \[\ce{P ->[Na2Cr2O7][H2SO4] R}\]
Q and R are respectively:
Which following reagent is used to obtain alkene from alcohol?
Which among the following compounds is used to prepare solid fuel using calcium acetate?
The number of σ bonds in carbolic acid is ______.
Which of the following compounds does NOT react with sodium metal?
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
A reaction of phenol with chloroform in presence of sodium hydroxide to form salicylaldehyde is known as ____________.
The chemical test that distinguish between benzoic acid and phenol is ______.
The order of reactivity of hydrogen halides with ether is as follows.
The major product obtained in the following reaction is

Explain: Phenols are acid while alcohol is neutral.
What is the commercial method of preparation of phenol?
