Advertisements
Advertisements
Question
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
Advertisements
Solution
Ethyl alcohol gives hydrogen on reaction with sodium metal. On oxidation with PCC ethanol will form acetaldehyde (C2H4O).
\[\ce{\underset{\text{Ethyl alcohol}}{2CH3 - CH2 - OH} ->[2Na] \underset{\text{Sodium ethoxide}}{2CH3 - CH2 - O- Na+} + {H_{2(g)}} ^}\]
\[\ce{\underset{\text{Ethyl alcohol}}{CH3 - CH2 - OH} ->[{[O]}][PCC] \underset{\ce{Acetaldehyde(C2H4O)}}{CH3 - CHO} + H2O}\]
RELATED QUESTIONS
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
Reaction between Grignard reagent and aldehyde other than formaldehyde leads to formation of _______________
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
How will you bring about the following conversions?
isopropyl alcohol to acetone
Write chemical reactions to convert phenol into salicyaldehyde. Write the name of the reaction. What happens when if CCl4 is used instead of CHCl3 in the above reaction.
Phenol is obtained from cumene ____________.
Propane when treated with cold cone. H2SO4 forms a compound which on heating with water gives ______.
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
The product 'C' in the following reaction is:
\[\ce{CH3CH2Br ->[alc. KCN] {'A'} ->[H3O^+][\Delta] {'B'} ->[i. LiAlH4][ii. H3O^+] {'C'}}\]
Carbolic acid is oxidised by acidified sodium dichromate to give ______.
The most resistant alcohol towards oxidation reaction is:
Which isomer of C4H10O is optically active?
Which among the following is allylic secondary alcohol?
Identify 'Z' in the following series of reaction:
\[\ce{Butan - 2 - ol ->[PCl3] X ->[alco. KOH] Y ->[i) H2SO4][ii) H-OH/heat] Z}\]
The compound which reacts fastest with Lucas reagent at room temperature is ______.

Product (B) in this reaction is:
Which of the following compounds has lowest boiling point?
Which of the following compounds reacts immediately with Lucas reagent?
Arrange the following compounds in an increasing order of their solubility in water:
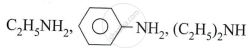
Write chemical reaction when hot copper is treated with Vapours of 3° (tertiary) alcohol.
What is the commercial method of preparation of phenol?
Arrange the following compounds in order of decreasing acidity:
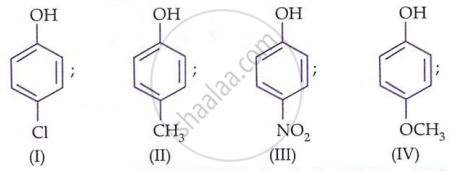
The structure of A in the given reaction is:
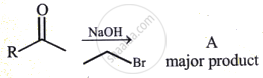
The C−O−C angle in ether is about ______.
Which of the following is primary benzylic alcohol?
Which of the following is trihydric phenol?
