Advertisements
Advertisements
प्रश्न
Derive Laplace’s law for spherical membrane of bubble due to surface tension.
Derive Laplace’s law for a spherical membrane.
Advertisements
उत्तर १
Consider a spherical liquid drop and let the outside pressure be Po and inside pressure be Pi, such that the excess pressure is Pi − Po

Let the radius of the drop increase from r to Δr, where Δr is very small, so that the pressure inside the drop remains almost constant.
Initial surface area (A1) = 4Πr2
Final surface area (A2) = 4Π(r + Δr)2
= 4π(r2 + 2rΔr + Δr2)
= 4Πr2 + 8ΠrΔr + 4ΠΔr2
As Δr is very small, Δr2 is neglected (i.e. 4πΔr2≅0)
Increase in surface area (dA) =A2 - A1= 4Πr2 + 8ΠrΔr - 4Πr2
Increase in surface area (dA) =8ΠΔr
Work done to increase the surface area by 8ΠrΔr is extra energy.
∴ dW = TdA
∴ dW = T*8πrΔr .......(Equ.1)
This work done is equal to the product of the force and the distance Δr.
dF=(P1 - P0)4πr2
The increase in the radius of the bubble is Δr.
dW = dFΔr = (P1 - P0)4Πr2*Δr ..........(Equ.2)
Comparing Equations 1 and 2, we get
(P1 - P0)4πr2*Δr = T*8πrΔr
∴`(P_1 - P_0) = (2T)/R`
This is called Laplace’s law of spherical membrane.
उत्तर २
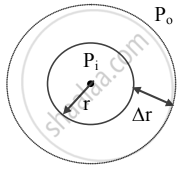
- The free surface of drops or bubbles is spherical in shape.
Let,
Pi = inside pressure of a drop or air bubble
Po = outside pressure of the bubble
r = radius of drop or bubble. - As drop is spherical, Pi > Po
∴ excess pressure inside drop = Pi − Po - Let the radius of drop increase from r to r + ∆r so that inside pressure remains constant.
- Initial area of drop A1 = 4πr2 ,
Final surface area of drop A2 = 4π (r + ∆r)2
Increase in surface area,
∆A = A2 − A1 = 4π[(r + ∆r)2 − r2]
= 4π[r2 + 2r∆r + ∆r2 − r2]
= 8πr∆r + 4π∆r2 - As ∆r is very small, the term containing ∆r2 can be neglected.
∴ dA = 8πr∆r - Work is done by a force of surface tension,
dW = TdA = (8πr∆r)T ….(1)
This work done is also equal to the product of the force F which causes an increase in the area of the bubble and the displacement Δr which is the increase in the radius of the bubble.
∴ dW = FΔr
The excess force is given by,
(Excess pressure) × (Surface area)
∴ F = (Pi – Po) × 4πr2
∴ dF = (Pi – Po)A
dW = F∆r = (Pi − Po) A∆r
From equation (1),
(Pi − Po) A∆r = (8πr∆r) T
∴ Pi − Po = `(8pirDeltarT)/(4pir^2Deltar)` ........(∵ A = 4πr2)
∴ Pi − Po = `(2T)/r` ….(2)
Equation (2) represents Laplace’s law of spherical membrane.
APPEARS IN
संबंधित प्रश्न
Angle of contact for the pair of pure water with clean glass is _______.
A raindrop of diameter 4 mm is about to fall on the ground. Calculate the pressure inside the raindrop. [Surface tension of water T = 0.072 N/m, atmospheric pressure = 1.013 x 105 N/m2 ]
'n' droplets of equal size of radius r coalesce to form a bigger drop of radius R. The energy liberated is equal to...................
(T =Surface tension of water)
`(a) 4piR^2T[n^(1/3)-1]`
`(b) 4pir^2T[n^(1/3)-1]`
`(c) 4piR^2T[n^(2/3)-1]`
`(d)4 pir^2T[n^(2/3)-1]`
Explain why A drop of liquid under no external forces is always spherical in shape
Mercury has an angle of contact equal to 140° with soda lime glass. A narrow tube of radius 1.00 mm made of this glass is dipped in a trough containing mercury. By what amount does the mercury dip down in the tube relative to the liquid surface outside? Surface tension of mercury at the temperature of the experiment is 0.465 N m–1. Density of mercury = 13.6 × 103 kg m–3
Define surface tension and surface energy.
In a conical pendulum, a string of length 120 cm is fixed at rigid support and carries a mass
of 150 g at its free end. If the mass is revolved in a horizontal circle of radius 0.2 m around a
vertical axis, calculate tension in the string (g = 9.8 m/s2)
State any two characteristics of the angle of contact
When the size of a soap bubble is increased by pushing more air in it, the surface area increases. Does it mean that the average separation between the surface molecules is increased?
Water near the bed of a deep river is quiet while that near the surface flows. Give reasons.
If water in one flask and castor oil in other are violently shaken and kept on a table, which will come to rest earlier?
By a surface of a liquid we mean
An ice cube is suspended in vacuum in a gravity free hall. As the ice melts it
When water droplets merge to form a bigger drop
If more air is pushed in a soap bubble, the pressure in it
The excess pressure inside a soap bubble is twice the excess pressure inside a second soap bubble. The volume of the first bubble is n times the volume of the second where n is
Water rises in a vertical capillary tube up to a length of 10 cm. If the tube is inclined at 45°, the length of water risen in the tube will be
The rise of a liquid in a capillary tube depends on
(a) the material
(b) the length
(c) the outer radius
(d) the inner radius of the tube
The contact angle between a solid and a liquid is a property of
(a) the material of the solid
(b) the material of the liquid
(c) the shape of the solid
(d) the mass of the solid
A liquid is contained in a vertical tube of semicircular cross section. The contact angle is zero. The force of surface tension on the curved part and on the flat part are in ratio

When a capillary tube is dipped into a liquid, the liquid neither rises nor falls in the capillary.
(a) The surface tension of the liquid must be zero.
(b) The contact angle must be 90°.
(c) The surface tension may be zero.
(d) The contact angle may be 90°.
Find the excess pressure inside (a) a drop of mercury of radius 2 mm (b) a soap bubble of radius 4 mm and (c) an air bubble of radius 4 mm formed inside a tank of water. Surface tension of mercury, soap solution and water are 0.465 N m−1, 0.03 N m−1 and 0.076 N m−1 respectively.
A capillary tube of radius 0.50 mm is dipped vertically in a pot of water. Find the difference between the pressure of the water in the tube 5.0 cm below the surface and the atmospheric pressure. Surface tension of water = 0.075 N m−1.
The lower end of a capillary tube of radius 1 mm is dipped vertically into mercury. (a) Find the depression of mercury column in the capillary. (b) If the length dipped inside is half the answer of part (a), find the angle made by the mercury surface at the end of the capillary with the vertical. Surface tension of mercury = 0.465 N m−1 and the contact angle of mercury with glass −135 °.
Two large glass plates are placed vertically and parallel to each other inside a tank of water with separation between the plates equal to 1 mm. Find the rise of water in the space between the plates. Surface tension of water = 0.075 Nm−1.
A ferry boat has internal volume 1 m3 and weight 50 kg.(a) Neglecting the thickness of the wood, find the fraction of the volume of the boat immersed in water.(b) If a leak develops in the bottom and water starts coming in, what fraction of the boat's volume will be filled with water before water starts coming in from the sides?
A cube of ice floats partly in water and partly in K.oil (in the following figure). Find the ratio of the volume of ice immersed in water to that in K.oil. Specific gravity of K.oil is 0.8 and that of ice is 0.9.

A cubical box is to be constructed with iron sheets 1 mm in thickness. What can be the minimum value of the external edge so that the cube does not sink in water? Density of iron = 8000 kg/m3 and density of water = 1000 kg/m3.
A cubical metal block of edge 12 cm floats in mercury with one fifth of the height inside the mercury. Water in it. Find the height of the water column to be poured.
Specific gravity of mercury = 13.6.
A solid sphere of radius 5 cm floats in water. If a maximum load of 0.1 kg can be put on it without wetting the load, find the specific gravity of the material of the sphere.
Water level is maintained in a cylindrical vessel up to a fixed height H. The vessel is kept on a horizontal plane. At what height above the bottom should a hole be made in the vessel so that the water stream coming out of the hole strikes the horizontal plane at the greatest distance from the vessel.

The energy stored in a soap bubble of diameter 6 cm and T = 0.04 N/m is nearly ______.
Calculate the rise of water inside a clean glass capillary tube of radius 0.1 mm, when immersed in water of surface tension 7 × 10-2 N/m. The angle of contact between water and glass is zero, the density of water = 1000 kg/m3, g = 9.8 m/s2.
Insect moves over the surface of water because of ______.
The water droplets are spherical in free fall due to ______
Define surface tension.
Water rises to a height of 20 mm in a capillary tube. If the radius made 1/3rd of its previous value, to what height will the water now rise in the tube?
How does the friction arise between the surfaces of two bodies in relative motion?
What do you mean by capillarity or capillary action?
What is capillarity?
Obtain an expression for the surface tension of a liquid by the capillary rise method.
The surface tension of the two liquids is respectively 20 and 60 dyne cm-1. The liquids drop from the ends of two tubes of the same radius. The ratio of the weights of the two drops is ______
Two spherical rain drops reach the surface of the earth with terminal velocities having ratio 16 : 9. The ratio of their surface area is ______.
A large number of liquid drops each of radius 'r' coalesce to form a big drop of radius 'R'. The energy released in the process in converted into kinetic energy of the big drop. The speed of the big drop is ______. (T = surface tension of liquid, p = density of liquid)
The wear and tear in the machine part is due to ______.
The length of a needle floating on water is 2 cm. The additional force due to surface tension required to pull the needle out of water will be (S.T. of water = 7.0 × 10−2 N/m).
The sap in trees, which consists mainly of water in summer, rises in a system of capillaries of radius r = 2.5 × 10–5 m. The surface tension of sap is T = 7.28 × 10–2 Nm–1 and the angle of contact is 0°. Does surface tension alone account for the supply of water to the top of all trees?
If a drop of liquid breaks into smaller droplets, it results in lowering of temperature of the droplets. Let a drop of radius R, break into N small droplets each of radius r. Estimate the drop in temperature.
This model of the atmosphere works for relatively small distances. Identify the underlying assumption that limits the model.
A liquid flows out drop by drop from a vessel through a vertical tube with an internal diameter of 2 mm, then the total number of drops that flows out during 10 grams of the liquid flow out ______. [Assume that the diameter of the neck of a drop at the moment it breaks away is equal to the internal diameter of tube and surface tension is 0.02 N/m].
In a U-tube, the radii of two columns are respectively r1 and r2. When a liquid of density ρ(θ = 0°) is filled in it, a level difference of h is observed on two arms, then the surface tension of the liquid is ______.
The surface tension of a soap solution is T. The work done in blowing a soap bubble of diameter d to that of a diameter 2d is ______.
Work done to blow a bubble of volume V is W. The work done in blowing a bubble of volume 2V will be ______.
The surface tension of soap solution is 25 × 10-3 Nm-1. The excess of pressure inside a soap bubble of diameter 1 cm is ______.
