Advertisements
Advertisements
प्रश्न
A ferry boat has internal volume 1 m3 and weight 50 kg.(a) Neglecting the thickness of the wood, find the fraction of the volume of the boat immersed in water.(b) If a leak develops in the bottom and water starts coming in, what fraction of the boat's volume will be filled with water before water starts coming in from the sides?
Advertisements
उत्तर
Internal volume, V = 1 m3 = External volume of the ferry boat
Density of water, \[\rho_w\] = 103 kg/m3
As the weight of the boat is balanced by the buoyant force, we have:
\[\text{mg} = V_1 \times \rho_w \times g\]
\[ \Rightarrow 50 = V_1 \times {10}^3 \]
\[ \Rightarrow V_1 = \frac{5}{100} = 0 . 05 \text{ m}^3\]
(b) Let V2 be the volume of the boat filled with water before water starts coming in from the side.
\[\therefore \text {mg + V}_2 \rho_\text{w} \times \text{g = V} \times \rho_\text{w} \times g [\text{ V is the volume of the water displaced by the boat }. ]\]
\[ \Rightarrow 50 + V_2 \times {10}^3 = 1 \times {10}^3 \]
\[ \Rightarrow V_2 = \frac{{10}^3 - 50}{{10}^3}\]
\[ = \frac{950}{1000} = 0 . 95 \text{ m}^3 \]
Fraction of the boat's volume filled with water\[ = \frac{19}{20}\]
APPEARS IN
संबंधित प्रश्न
The energy of the free surface of a liquid drop is 5π times the surface tension of the liquid. Find the diameter of the drop in C.G.S. system.
Derive an expression for excess pressure inside a drop of liquid.
Explain why Surface tension of a liquid is independent of the area of the surface
Explain why Water with detergent dissolved in it should have small angles of contact.
In a conical pendulum, a string of length 120 cm is fixed at rigid support and carries a mass
of 150 g at its free end. If the mass is revolved in a horizontal circle of radius 0.2 m around a
vertical axis, calculate tension in the string (g = 9.8 m/s2)
A liquid is contained in a vertical tube of semicircular cross section. The contact angle is zero. The force of surface tension on the curved part and on the flat part are in ratio

Find the force exerted by the water on a 2 m2 plane surface of a large stone placed at the bottom of a sea 500 m deep. Does the force depend on the orientation of the surface?
A cubical block of ice floating in water has to support a metal piece weighing 0.5 kg. Water can be the minimum edge of the block so that it does not sink in water? Specific gravity of ice = 0.9.
A cube of ice floats partly in water and partly in K.oil (in the following figure). Find the ratio of the volume of ice immersed in water to that in K.oil. Specific gravity of K.oil is 0.8 and that of ice is 0.9.

Derive an expression for capillary rise for a liquid having a concave meniscus.
Mention the S.I unit and dimension of surface tension.
How is surface tension related to surface energy?
Obtain an expression for the surface tension of a liquid by the capillary rise method.
Why coffee runs up into a sugar lump (a small cube of sugar) when one corner of the sugar lump is held in the liquid?
Two mercury droplets of radii 0.1 cm. and 0.2 cm. collapse into one single drop. What amount of energy is released? The surface tension of mercury T = 435.5 × 10–3 Nm–1.
A soap bubble of radius 3 cm is formed inside another soap bubble of radius 6 cm. The radius of an equivalent soap bubble which has the same excess pressure as inside the smaller bubble with respect to the atmospheric pressure is ______ cm.
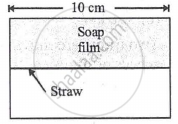
A soap film of surface tension 3 × 10-2 formed in a rectangular frame can support a straw as shown in Fig. If g = 10 ms-12, the mass of the straw is ______.
Work done to blow a bubble of volume V is W. The work done in blowing a bubble of volume 2V will be ______.
In most liquids, with the rise in temperature, the surface tension of a liquid ______.
Find the work done when a drop of mercury of radius 2 mm breaks into 8 equal droplets. [Surface tension of mercury = 0.4855 J/m2].
