Advertisements
Advertisements
प्रश्न
A beam of white light is incident normally on a plane surface absorbing 70% of the light and reflecting the rest. If the incident beam carries 10 W of power, find the force exerted by it on the surface.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Advertisements
उत्तर
Power of the incident beam, P = 10 watt
Relation between wavelength (λ) and momentum (p):
`λ = h/p` ,
where h is Planck's constant
⇒ `p = h/λ`
On dividing both sides by t , we get :
`p/t = h/(λt)` ...(1)
Energy,
`E = (hc)/λ`
⇒ `E/t = (hc)/(λt)`
Let P be the power . Then,
`P = E/t = (hc)/(λt)`
`P = (pc)/t ................["Using equation (1)"]`
⇒` p/c = p/t`
Force ,
`F = p/t = p/c` `("Since F" = "Momentum"/"Time")`
`"Force, F" = 7/10 " (absorted)" + 2 xx 3/10 ("reflected")`
`F = 7/10 xx P/c + 2 xx 3/10 xx P/c`
`F = 7/10 xx 10/(3 xx 10^8) + 2 xx 3/10 xx 10/(3 xx 10^8)`
`F = 13/3 xx 10^-8 = 4.33 xx 10^-8 "N"`
APPEARS IN
संबंधित प्रश्न
Define the term 'intensity of radiation' in terms of photon picture of light.
Ultraviolet light of wavelength 2271 Å from a 100 W mercury source irradiates a photo-cell made of molybdenum metal. If the stopping potential is −1.3 V, estimate the work function of the metal. How would the photo-cell respond to a high intensity (∼105 W m−2) red light of wavelength 6328 Å produced by a He-Ne laser?
The following graph shows the variation of photocurrent for a photosensitive metal :
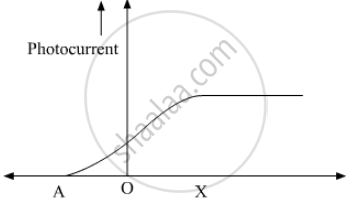
(a) Identify the variable X on the horizontal axis.
(b) What does the point A on the horizontal axis represent?
(c) Draw this graph for three different values of frequencies of incident radiation v1, v2 and v3 (v1 > v2 > v3) for same intensity.
(d) Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I1 > I2 > I3) having same frequency.
Is it always true that for two sources of equal intensity, the number of photons emitted in a given time are equal?
What is the speed of a photon with respect to another photon if (a) the two photons are going in the same direction and (b) they are going in opposite directions?
Should the energy of a photon be called its kinetic energy or its internal energy?
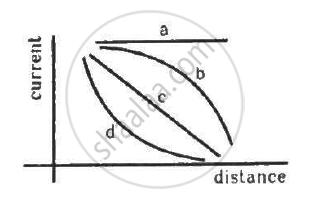
A point source causes photoelectric effect from a small metal plate. Which of the following curves may represent the saturation photocurrent as a function of the distance between the source and the metal?
In which of the following situations, the heavier of the two particles has smaller de Broglie wavelength? The two particles
(a) move with the same speed
(b) move with the same linear momentum
(c) move with the same kinetic energy
(d) have fallen through the same height
Calculate the momentum of a photon of light of wavelength 500 nm.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A 100 W light bulb is placed at the centre of a spherical chamber of radius 20 cm. Assume that 60% of the energy supplied to the bulb is converted into light and that the surface of the chamber is perfectly absorbing. Find the pressure exerted by the light on the surface of the chamber.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A totally reflecting, small plane mirror placed horizontally faces a parallel beam of light, as shown in the figure. The mass of the mirror is 20 g. Assume that there is no absorption in the lens and that 30% of the light emitted by the source goes through the lens. Find the power of the source needed to support the weight of the mirror.

(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The work function of a metal is 2.5 × 10−19 J. (a) Find the threshold frequency for photoelectric emission. (b) If the metal is exposed to a light beam of frequency 6.0 × 1014 Hz, what will be the stopping potential?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
When a metal plate is exposed to a monochromatic beam of light of wavelength 400 nm, a negative potential of 1.1 V is needed to stop the photo current. Find the threshold wavelength for the metal.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Why it is the frequency and not the intensity of the light source that determines whether the emission of photoelectrons will occur or not? Explain.
Read the following paragraph and answer the questions.
| The figure shows the variation of photoelectric current measured in a photocell circuit as a function of the potential difference between the plates of the photocell when light beams A, B, C and D of different wavelengths are incident on the photocell. Examine the given figure and answer the following questions: |

- Which light beam has the highest frequency and why?
- Which light beam has the longest wavelength and why?
- Which light beam ejects photoelectrons with maximum momentum and why?
How would the stopping potential for a given photosensitive surface change if the intensity of incident radiation was decreased? Justify your answer.
Plot a graph showing the variation of photoelectric current, as a function of anode potential for two light beams having the same frequency but different intensities I1 and I2 (I1 > I2). Mention its important features.
A metallic plate exposed to white light emits electrons. For which of the following colours of light, the stopping potential will be maximum?
The difference between threshold wavelengths for two metal surfaces A and B having work function ΦA = 9 eV and ΦB = 4.5 eV in nm is ______.
(Given, hc = 1242 eV nm)
