Advertisements
Advertisements
प्रश्न
When the sun is directly overhead, the surface of the earth receives 1.4 × 103 W m−2 of sunlight. Assume that the light is monochromatic with average wavelength 500 nm and that no light is absorbed in between the sun and the earth's surface. The distance between the sun and the earth is 1.5 × 1011 m. (a) Calculate the number of photons falling per second on each square metre of earth's surface directly below the sun. (b) How many photons are there in each cubic metre near the earth's surface at any instant? (c) How many photons does the sun emit per second?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Advertisements
उत्तर
Here,
Intensity of light, `l = 1.4 × 10^3` W/m2,
Wavelength of light, `λ = 500 "nm" = 500 xx 10^-9 "m"`
Distance between the Sun and Earth, `l = 1.5 xx 10^11 "m"`
Intensity,
`I = "power"/"Area" = 1.4 xx 10^3 "W/m"^2`
Let n be the number of photons emitted per second.
∴ Power, P = Energy emitted/second
`P = (nhc)/λ`,
where λ = wavelength of light
h = Planck's constant
c = speed of light
Number of photons/`"m"^2` = `(nhc)/(λ xx A) = (nhc)/(λ xx 1) = l`
`therefore n = (I xx λ)/(hc)`
= `(1.4 xx 10^3 xx 500 xx 10^-9)/(6.63 xx 10^-34 xx 3 xx 10^8)`
= `3.5 xx 10^21`
(b) Consider number of two parts at a distance r and r + dr from the source.
Let dt' be the time interval in which the photon travels from one part to another.
Total number of photons emitted in this time interval,
`N = ndt = ((Pλ)/(hc xx A))(dr)/c`
These points will be between two spherical shells of radius 'r' and r + dr. It will be the distance of the 1st point from the sources.
In this case,
`l = 1.5 xx 10^11 "m"`
`"Wavelength" , λ = 500 "nm" = 500 xx 10^-9 "m"`
`P/(4pir^2) = 1.4 xx 10^3`
∴ No. of photons `/"m"^3 = P/(4pir^2) λ/(hc^2)`
= `1.4 xx 10^3 xx (500 xx 10^-9)/(6.63 xx 10^-34 xx 9 xx 10^16`
= `1.2 xx 10^13`
(c) Number of photons emitted = (Number of photons / s-m2) × Area
= `(3.5 xx 10^21) xx 4pil^2`
= `3.5 xx 10^21 xx 4 xx (3.14) xx (1.5 xx 10^11)^2`
= `9.9 xx 10^44`
APPEARS IN
संबंधित प्रश्न
Define the term 'intensity of radiation' in terms of photon picture of light.
Use the same formula you employ in (a) to obtain electron speed for an collector potential of 10 MV. Do you see what is wrong? In what way is the formula to be modified?
Ultraviolet light of wavelength 2271 Å from a 100 W mercury source irradiates a photo-cell made of molybdenum metal. If the stopping potential is −1.3 V, estimate the work function of the metal. How would the photo-cell respond to a high intensity (∼105 W m−2) red light of wavelength 6328 Å produced by a He-Ne laser?
Monochromatic radiation of wavelength 640.2 nm (1 nm = 10−9 m) from a neon lamp irradiates photosensitive material made of caesium on tungsten. The stopping voltage is measured to be 0.54 V. The source is replaced by an iron source and its 427.2 nm line irradiates the same photo-cell. Predict the new stopping voltage.
Every metal has a definite work function. Why do all photoelectrons not come out with the same energy if incident radiation is monochromatic? Why is there an energy distribution of photoelectrons?
Can we find the mass of a photon by the definition p = mv?
What is the speed of a photon with respect to another photon if (a) the two photons are going in the same direction and (b) they are going in opposite directions?
It is found that yellow light does not eject photoelectrons from a metal. Is it advisable to try with orange light or with green light?
Planck's constant has the same dimensions as
A point source of light is used in a photoelectric effect. If the source is removed farther from the emitting metal, the stopping potential
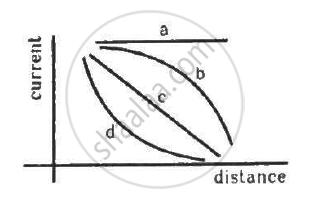
A point source causes photoelectric effect from a small metal plate. Which of the following curves may represent the saturation photocurrent as a function of the distance between the source and the metal?
Calculate the number of photons emitted per second by a 10 W sodium vapour lamp. Assume that 60% of the consumed energy is converted into light. Wavelength of sodium light = 590 nm
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, find the force exerted by the light beam on the sphere.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Answer the following question.
Plot a graph of photocurrent versus anode potential for radiation of frequency ν and intensities I1 and I2 (I1 < I2).
Explain how does (i) photoelectric current and (ii) kinetic energy of the photoelectrons emitted in a photocell vary if the frequency of incident radiation is doubled, but keeping the intensity same?
Show the graphical variation in the above two cases.
In photoelectric effect the photo current ______.
How would the stopping potential for a given photosensitive surface change if the frequency of the incident radiation were increased? Justify your answer.
The difference between threshold wavelengths for two metal surfaces A and B having work function ΦA = 9 eV and ΦB = 4.5 eV in nm is ______.
(Given, hc = 1242 eV nm)
