Topics
Solutions
- Introduction to Solution
- Expressing Concentration of Solutions
- Solubility
- Solubility of a Solid in a Liquid
- Solubility of a Gas in a Liquid
- Vapour Pressure of Liquid Solutions
- Vapour Pressure of Liquid- Liquid Solutions
- Raoult’s Law as a Special Case of Henry’s Law
- Vapour Pressure of Solutions of Solids in Liquids
- Ideal and Non-ideal Solutions
- Colligative Properties and Determination of Molar Mass
- Relative Lowering of Vapour Pressure
- Elevation of Boiling Point
- Depression of Freezing Point
- Osmosis and Osmotic Pressure
- Reverse Osmosis and Water Purification
- Abnormal Molar Masses
- Solution Numericals
- Quantitative Concentration Numericals
- Composition of Vapour Phase
- Overview of Solutions
- Kohlrausch's law
Solid State
- General Characteristics of Solid State
- Amorphous and Crystalline Solids
- Classification of Crystalline Solids
- Crystal Lattices and Unit Cells
- Crystal Lattices and Unit Cells - Primitive and Centred Unit Cells
- Number of Atoms in a Unit Cell
- Close Packed Structures of Solids
- Close Packed Structures - Formula of a Compound and Number of Voids Filled
- Packing Efficiency in hcp and ccp Structures
- Efficiency of Packing in Body-centred Cubic Structures
- Packing Efficiency in Simple Cubic Lattice
- Calculations Involving Unit Cell Dimensions
- Imperfections in Solids - Introduction
- Types of Point Defects - Stoichiometric Defects
- Types of Point Defects - Impurity Defects
- Types of Point Defects - Non-stoichiometric Defects
- Properties of Solids: Electrical Properties
- Conduction of Electricity in Metals
- Conduction of Electricity in Semiconductors
- Applications of n-type and p-type Semiconductors
- Properties of Solids: Magnetic Properties
- Band Theory of Metals
- Solid State Numericals
Electrochemistry
- Introduction to Electrochemistry
- Electrochemical Cells
- Galvanic or Voltaic Cell
- Galvanic Cells - Measurement of Electrode Potential
- Nernst Equation
- Equilibrium Constant from Nernst Equation
- Electrochemical Cell and Gibbs Energy of the Reaction
- Conductance of Electrolytic Solutions - Introduction
- Measurement of the Conductivity of Ionic Solutions
- Variation of Conductivity and Molar Conductivity with Concentration
- Electrolytic Cells and Electrolysis - Introduction
- Products of Electrolysis
- Primary Batteries
- Secondary Batteries
- Fuel Cells
- Corrosion of Metals
- Relation Between Gibbs Energy Change and Emf of a Cell
- Lead Accumulator
- Faraday's Laws of Electromagnetic Induction
- Overview of Electrochemistry
Chemical Kinetics
- Rate of Chemical Reaction
- Factors Influencing Rate of a Reaction
- Integrated Rate Equations
- Zero Order Reactions
- First Order Reactions
- Half Life Period of a Reaction
- Pseudo First Order Reaction
- Temperature Dependence of the Rate of a Reaction
- Collision Theory of Chemical Reactions
- Effect of Catalyst on the Rate of Reaction
- Kinetic Energy of Molecule
- Role of Catalyst
- Rate Law and Specific Rate Constant
- Overview of Chemical Kinetics
d-block and f-block Elements
- General Introduction of "d" and "f" Block Element
- Position in the Periodic Table - d-block Elements
- Electronic Configurations of the d-block Elements
- General Properties of the Transition Elements (d-block)
- Important Compounds of Transition Elements
- The Lanthanoids
- The Actinoids
- Applications of d and f Block Elements
- "D" and "F" Block Elements Numericals
- Overview of d-block and f-block Elements
Coordination Compounds
- Concept of Coordination Compounds
- Werner’s Theory of Coordination Compounds
- Important Terms Pertaining to Coordination Compounds
- Types of Ligands
- Nomenclature of Coordination Compounds
- Isomerism in Coordination Compounds
- Stereoisomerism
- Structural Isomerism
- Bonding in Coordination Compounds
- Valence Bond Theory (VBT)
- Magnetic Properties of Coordination Compounds
- Crystal Field Theory (CFT)
- Colour in Coordination Compounds
- Bonding in Metal Carbonyls
- Stability of Coordination Compounds
- Importance and Applications of Coordination Compounds
- Coordination Compounds Numerical
- Overview of Coordination Compounds
Surface Chemistry
- Introduction of Adsorption
- Distinction Between Adsorption and Absorption
- Mechanism of Adsorption
- Types of Adsorption
- Adsorption Isotherms (Freundlich and Langmuir Adsorption Isotherm)
- Adsorption from Solution Phase
- Applications of Adsorption
- Homogeneous and Heterogeneous Catalysis
- Adsorption Theory of Heterogeneous Catalysis
- Shape-selective Catalysis by Zeolites
- Enzyme Catalysis
- Catalysts in Industry
- Colloids
- Preparation of Colloids
- Purification of Colloidal Solution
- Properties of Colloidal Dispersions
- Preparation of Colloids
- Purification of Colloidal Solution
- Properties of Colloidal Solutions
- Emulsions
- Colloids Around Us
Haloalkanes and Haloarenes
- Introduction to Haloalkanes and Haloarenes
- Classification of Haloalkanes and Haloarenes
- Nomenclature
- Nature of C-X Bond
- Methods of Preparation of Haloalkanes
- Methods of Preparation of Haloarenes
- Physical Properties of Haloalkanes and Haloarenes
- Reactions of Haloalkanes - Nucleophilic Substitution Reactions
- Reactions of Haloalkanes - Elimination Reactions
- Reactions of Haloalkanes - Reaction with Metals
- Reactions of Haloarenes - Nucleophilic Substitution
- Reactions of Haloarenes - Electrophilic Substitution Reactions
- Reactions of Haloarenes - Reaction with Metals
- Polyhalogen Compounds
- R-s and D-l Configuration
- Haloalkanes and Haloarenes Numericals
- Overview of Haloalkanes and Haloarenes
General Principles and Processes of Isolation of Elements
- Occurrence of Metals
- Types of Separation or Concentration of an Ore
- Hydraulic Washing
- Magnetic Separation
- Froth Floatation Method
- Leaching
- Extraction of Crude Metal from Concentrated Ore
- Thermodynamic Principles of Metallurgy
- Application of Thermodynamic Principles of Metallurgy
- Electrochemical Principles of Metallurgy
- Oxidation Reduction
- Refining of Crude Metals
- Principles and Methods of Extraction - Concentration
- Uses of Aluminium, Copper, Zinc and Iron
- General Principles and Processes of Isolation of Elements Numerical
P - Block Elements
- Group 15 Elements - The Nitrogen Family
- Dinitrogen
- Ammonia
- Oxides of Nitrogen
- Nitric Acid
- Phosphorus - Allotropic Forms
- Compounds of Phosphorus
- Phosphine
- Phosphorus Halides
- Oxoacids of Phosphorus
- Group 16 Elements - The Oxygen Family
- Dioxygen
- Classification of Oxides
- Simple Oxides
- Ozone
- Sulphur - Allotropic Forms
- Compounds of Sulphur
- Sulphur Dioxide
- Oxoacids of Sulphur
- Sulphuric Acid
- Group 17 Elements - The Halogen Family
- Compounds of Halogens
- Chlorine
- Hydrogen Chloride
- Oxoacids of Halogens
- Interhalogen Compounds
- Group 18 Elements - The Noble gas Family
- P Block Elements
Alcohols, Phenols and Ethers
- Classification of Alcohols and Phenols
- Classification of Ethers
- Nomenclature
- Structures of Functional Groups of Alcohols, Phenols and Ethers
- Methods of Preparation of Alcohols
- Methods of Preparation of Phenols
- Chemical Properties of Alcohols and Phenols
- Reactions Involving Cleavage of O-H Bond
- Reactions Involving Cleavage of Carbon–Oxygen (C–O) Bond in Alcohols
- Chemical Properties of Phenol
- Preparation of Commercially Important Alcohols
- Preparation of Ethers
- Physical Properties of Ethers
- Chemical Reaction of Ethers - Cleavege of C-O Bonds
- Chemical Reaction of Ethers - Electrophilic Substitution
- Overview of Alcohols, Phenols and Ethers
Aldehydes, Ketones and Carboxylic Acids
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Nomenclature of Aldehydes and Ketones
- Nature of Carbonyl Group
- Structure of the Carbonyl Group
- Preparation of Aldehydes and Ketones
- Preparation of Aldehydes
- Preparation of Ketones
- Physical Properties of Aldehydes and Ketones
- Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
- Chemical Reactions of Aldehydes and Ketones - Reduction
- Chemical Reactions of Aldehydes and Ketones - Oxidation
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Chemical Reactions of Aldehydes and Ketones - Other Reactions
- Uses of Aldehydes and Ketones
- Carboxylic Acids
- Nomenclature of Carboxylic Acids
- Structure of the Carboxyl group
- Methods of Preparation of Carboxylic Acids
- Physical Properties of Carboxylic Acids
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
- Chemical Reactions of Carboxylic Acids - Reactions Involving –COOH Group
- Chemical Reactions of Carboxylic Acids - Substitution Reactions in the Hydrocarbon Part
- Uses of Carboxylic Acids
- Overview: Aldehydes, Ketones and Carboxylic Acids
Amines
- Concept of Amines
- Structure of Amines
- Classification of Amines
- Nomenclature of Animes
- Preparation of Amines
- Physical Properties of Amines
- Chemical Reactions of Amines - Basic Character of Amines
- Chemical Reactions of Amines - Alkylation and Acylation
- Chemical Reactions of Amines - Carbylamine Reaction
- Chemical Reactions of Amines - Reaction with Nitrous Acid
- Chemical Reactions of Amines - Reaction with Arylsulphonyl Chloride
- Chemical Reactions of Amines - Electrophilic Substitution
- Uses of Amines
- Identification of Primary, Secondary and Tertiary Amines
- Cyanides and Isocyanides
- Diazonium Salts
- Method of Preparation of Diazonium Salts
- Physical Properties of Diazonium Salts
- Chemical Reaction of Diazonium Salts - Reactions Involving Displacement of Nitrogen
- Chemical Reaction of Diazonium Salts - Reactions Involving Retention of Diazo Group
- Importance of Diazonium Salts in Synthesis of Aromatic Compounds
- Organic Compounds Containing Nitrogen Numericals
- Overview of Amines
Biomolecules
- Biomolecules in the Cell > Carbohydrates
- Classification of Carbohydrates
- Monosaccahrides
- Preparation of Glucose
- Structures of Glucose
- Fructose
- Disaccharides
- Polysaccharides
- Oligosaccharides
- Polysaccharides
- Importance of Carbohydrates
- Introduction of Proteins
- Amino Acids
- Classification of Amino Acids
- Structure of Proteins
- Denaturation of Proteins
- Peptide
- Biomolecules in the Cell > Enzymes
- Mechanism of Enzyme Action
- Biomolecules in the Cell > Proteins
- Classification of Vitamins
- Biomolecules in the Cell > Nucleic Acids
- Chemical Composition of Nucleic Acids
- Structure of Nucleic Acids
- Biological Functions of Nucleic Acids
- Biomolecules in the Cell > Hormones
- Biomolecules Numericals
- Chemical Coordination
- Overview of Biomolecules
Polymers
- Introduction to Polymers
- Classification of Polymers> Based on Source or Origin
- Classification of Polymers> Based on Structure
- Classification of Polymers> Based on Mode of Polymerisation
- Classification of Polymers> Based on Intermolecular Forces
- Classification of Polymers Based on Growth Polymerisation
- Types of Polymerisation Reactions - Addition Polymerisation or Chain Growth Polymerisation
- Types of Polymerisation Reactions - Condensation Polymerisation Or Step Growth Polymerisation
- Types of Polymerisation Reactions - Copolymerisation
- Types of Polymerisation Reactions - Rubber
- Molecular Mass of Polymers
- Classification of Polymers> Based on Biodegradability
- Polymers of Commercial Importance
- Some Important Polymers
- Polymers Numericals
Chemistry in Everyday Life
- Classification of Drugs
- Drug-target Interaction - Enzymes as Drug Targets
- Drug-target Interaction - Receptors as Drug Targets
- Therapeutic Action of Different Classes of Drugs - Antacids
- Therapeutic Action of Different Classes of Drugs - Antihistamines
- Therapeutic Action of Different Classes of Drugs - Neurologically Active Drugs
- Antimicrobials
- Therapeutic Action of Different Classes of Drugs - Antifertility Drugs
- Chemicals in Food - Artificial Sweetening Agents and Food Preservatives
- Cleansing Agents
- Chemistry in Everyday Life Numericals
Estimated time: 10 minutes
CBSE: Class 12
Key Points: Important Compounds of Transition Elements
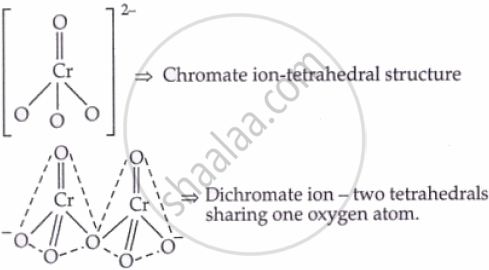
I. K₂Cr₂O₇ (Potassium dichromate)
Preparation: From chromite (FeO·Cr₂O₃)
4FeO·Cr₂O₃ + O₂ → 2Fe₂O₃ + 4Cr₂O₃
4Na₂CO₃ + 2Cr₂O₃ + 3O₂ → 4Na₂Cr₂O₄ + 4CO₂ × 2
4FeO·Cr₂O₃ + 8Na₂CO₃ + 7O₂ → 8Na₂Cr₂O₄ + 2Fe₂O₃ + 8CO₂
2Na₂CrO₄ + 8H₂SO₄ → Na₂Cr₂O₇ + Na₂SO₄ + H₂O
Na₂Cr₂O₇ + 2KCl → K₂Cr₂O₇ + 2NaCl
Properties
Oxidising Properties:
- Liberates I₂ from KI
K₂Cr₂O₇ + 7H₂SO₄ + 6KI → 4K₂SO₄ + Cr₂(SO₄)₃ + 3I₂ + 7H₂O - Oxidises ferrous salts to ferric salts:
K₂Cr₂O₇ + 7H₂SO₄ + 6FeSO₄ → K₂SO₄ + Cr₂(SO₄)₃ + 3Fe₂(SO₄)₃ + 2H₂O - Oxidises H₂S to sulphur:
K₂Cr₂O₇ + 4H₂SO₄ + 3H₂S → K₂SO₄ + Cr₂(SO₄)₃ + 7H₂O + 3S - Oxidises sulphites to sulphates:
K₂Cr₂O₇ + 4H₂SO₄ + 3Na₂SO₃ → K₂SO₄ + Cr₂(SO₄)₃ + 4H₂O + 3Na₂SO₄ - Oxidises nitrites to nitrates:
K₂Cr₂O₇ + 4H₂SO₄ + 3NaNO₂ → K₂SO₄ + Cr₂(SO₄)₃ + 3NaNO₃ + 4H₂O
Structures:
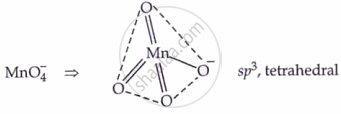
II. Potassium Permanganate (KMnO₄)
Preparation: From Pyrolusite (MnO₂)
2MnO₂ + 4KOH + O₂ → 2K₂MnO₄ + 2H₂O
2MnO₂ + 2K₂CO₃ + O₂ → 2K₂MnO₄ + 2CO₂
MnO₂ + 2KOH + KNO₃ → K₂MnO₄ + KNO₂ + H₂O
3K₂MnO₄ + 2CO₂ → 2KMnO₄ + MnO₂↓ + 2K₂CO₃
Properties
Oxidising Properties:
- Oxidising H₂S to S
2KMnO₄ + 3H₂SO₄ + 5H₂S → K₂SO₄ + 2MnSO₄ + 3H₂O + 5S - Oxidises sulphur dioxide to sulphuric acid
2KMnO₄ + 5SO₂ + 2H₂O → K₂SO₄ + 2MnSO₄ + 2H₂SO₄ - Oxidises oxalates to CO₂
2KMnO₄ + 3H₂SO₄ + 5C₂H₂O₄ → K₂SO₄ + 2MnSO₄ + 8H₂O + 10CO₂ - Oxidises HX to X₂, where X = Cl, Br, I
2KMnO₄ + 3H₂SO₄ + 10HX → K₂SO₄ + 2MnSO₄ + 8H₂O + 5X₂
Structure: