Advertisements
Advertisements
A primary alkyl halide would prefer to undergo ______.
Concept: undefined >> undefined

Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Concept: undefined >> undefined
Advertisements
Which of the following statements are correct about this reaction?

(i) The given reaction follows SN2 mechanism.
(ii) (b) and (d) have opposite configuration.
(iii) (b) and (d) have same configuration.
(iv) The given reaction follows SN1 mechanism.
Concept: undefined >> undefined
Which of the following statements are correct about the kinetics of this reaction?

(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
Concept: undefined >> undefined
Ethylene chloride and ethylidene chloride are isomers. Identify the correct statements.
(i) Both the compounds form same product on treatment with alcoholic KOH.
(ii) Both the compounds form same product on treatment with aq.NaOH.
(iii) Both the compounds form same product on reduction.
(iv) Both the compounds are optically active.
Concept: undefined >> undefined
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Concept: undefined >> undefined
Compound ‘A’ with molecular formula \[\ce{C4H9Br}\] is treated with aq. \[\ce{KOH}\] solution. The rate of this reaction depends upon the concentration of the compound ‘A’ only. When another optically active isomer ‘B’ of this compound was treated with aq. \[\ce{KOH}\] solution, the rate of reaction was found to be dependent on concentration of compound and \[\ce{KOH}\] both.
(i) Write down the structural formula of both compounds ‘A’ and ‘B’.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
Concept: undefined >> undefined
Write the structures and names of the compounds formed when compound ‘A’ with molecular formula, \[\ce{C7H8}\] is treated with \[\ce{Cl2}\] in the presence of \[\ce{FeCl3}\].
Concept: undefined >> undefined
How do polar solvents help in the first step in SN1 mechanism?
Concept: undefined >> undefined
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Concept: undefined >> undefined
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) |  |
| (II) |  |
| (III) |  |
Concept: undefined >> undefined
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
Concept: undefined >> undefined
Match the reactions given in Column I with the types of reactions given in Column II.
| Column I | Column II | |
| (i) | 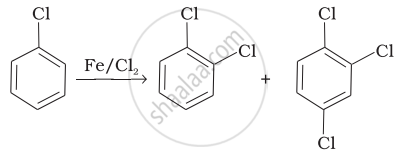 |
(a) Nucleophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(b) Electrophilic aromatic substitution |
| (iii) | 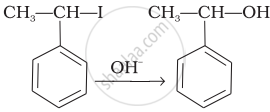 |
(c) Saytzeff elimination |
| (iv) | 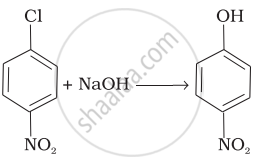 |
(d) Electrophilic addition |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(e) Nucleophilic substitution (SN1) |
Concept: undefined >> undefined
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides?
Concept: undefined >> undefined
Which of the following compounds will give butanone on oxidation with alkaline \[\ce{KMnO4}\] solution?
Concept: undefined >> undefined
Oxidation of ketones involves carbon-carbon bond cleavage. Name the products formed on oxidation of 2, 5-dimethylhexan-3-one.
Concept: undefined >> undefined
What is the role of pyridine in the acylation reaction of amines?
Concept: undefined >> undefined
Assertion: Acylation of amines gives a monosubstituted product whereas alkylation of amines gives polysubstituted product.
Reason: Acyl group sterically hinders the approach of further acyl groups.
Concept: undefined >> undefined
In disaccharides, if the reducing groups of monosaccharides i.e. aldehydic or ketonic groups are bonded, these are non-reducing sugars. Which of the following disaccharide is a non-reducing sugar?
Concept: undefined >> undefined
Match the following enzyms given in Column I with the reactions they catalyse given in Column II.
| Column I (Enzymes) | Column II (Reactions) |
| (i) Invertase | (a) Decomposition of urea into NH3 and CO2 |
| (ii) Maltase | (b) Conversion of glucose into ethyl alcohol |
| (iii) Pepsin | (c) Hydrolysis of maltose into glucose |
| (iv) Urease | (d) Hydrolysis of cane sugar |
| (v) Zymase | (e) Hydrolysis of proteins into peptides |
Concept: undefined >> undefined
