Advertisements
Advertisements
प्रश्न
The electric field associated with a light wave is given by `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`. Find the stopping potential when this light is used in an experiment on photoelectric effect with the emitter having work function 1.9 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Advertisements
उत्तर
Given :-
Electric field , `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`
Work function, `phi = 1.9 "eV"`
On comparing the given equation with the standard equation, `E = E_0 sin (kx - wt)`,
we get :-
`omega = 1.57 xx 10^7 xx c`
Now , frequency,
`v = (1.57 xx 10^7 xx 3 xx 10^8)/(2pi) Hz`
From Einstein's photoelectric equation,
`"eV"_0 = hv - phi`
Here, V0 = stopping potential
e = charge on electron
h = Planck's constant
On substituting the respective values, we get :-
`"eV"_0 = 6.63 xx 10^-34 xx (1.57 xx 3 xx 10^15)/(2pi xx 1.6 xx 10^-19) - 1.9 "eV"`
`⇒ "eV"_0 = 3.105 - 1.9 = 1.205 "eV"`
`⇒ V_0 = (1.205 xx 1.6 xx 10^-19)/(1.6 xx 10^-19) = 1.205 V`
Thus, the value of the stopping potential is 1.205 V.
APPEARS IN
संबंधित प्रश्न
Monochromatic radiation of wavelength 640.2 nm (1 nm = 10−9 m) from a neon lamp irradiates photosensitive material made of caesium on tungsten. The stopping voltage is measured to be 0.54 V. The source is replaced by an iron source and its 427.2 nm line irradiates the same photo-cell. Predict the new stopping voltage.
A mercury lamp is a convenient source for studying frequency dependence of photoelectric emission, since it gives a number of spectral lines ranging from the UV to the red end of the visible spectrum. In our experiment with rubidium photo-cell, the following lines from a mercury source were used:
λ1 = 3650 Å, λ2 = 4047 Å, λ3 = 4358 Å, λ4 = 5461 Å, λ5 = 6907 Å,
The stopping voltages, respectively, were measured to be:
V01 = 1.28 V, V02 = 0.95 V, V03 = 0.74 V, V04 = 0.16 V, V05 = 0 V
Determine the value of Planck’s constant h, the threshold frequency and work function for the material.
[Note: You will notice that to get h from the data, you will need to know e (which you can take to be 1.6 × 10−19 C). Experiments of this kind on Na, Li, K, etc. were performed by Millikan, who, using his own value of e (from the oil-drop experiment) confirmed Einstein’s photoelectric equation and at the same time gave an independent estimate of the value of h.]
Every metal has a definite work function. Why do all photoelectrons not come out with the same energy if incident radiation is monochromatic? Why is there an energy distribution of photoelectrons?
The following graph shows the variation of photocurrent for a photosensitive metal :
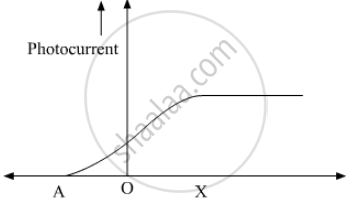
(a) Identify the variable X on the horizontal axis.
(b) What does the point A on the horizontal axis represent?
(c) Draw this graph for three different values of frequencies of incident radiation v1, v2 and v3 (v1 > v2 > v3) for same intensity.
(d) Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I1 > I2 > I3) having same frequency.
Draw graphs showing variation of photoelectric current with applied voltage for two incident radiations of equal frequency and different intensities. Mark the graph for the radiation of higher intensity.
Is it always true that for two sources of equal intensity, the number of photons emitted in a given time are equal?
What is the speed of a photon with respect to another photon if (a) the two photons are going in the same direction and (b) they are going in opposite directions?
It is found that yellow light does not eject photoelectrons from a metal. Is it advisable to try with orange light or with green light?
It is found that photosynthesis starts in certain plants when exposed to sunlight, but it does not start if the plants are exposed only to infrared light. Explain.
The work function of a metal is hv0. Light of frequency v falls on this metal. Photoelectric effect will take place only if
A totally reflecting, small plane mirror placed horizontally faces a parallel beam of light, as shown in the figure. The mass of the mirror is 20 g. Assume that there is no absorption in the lens and that 30% of the light emitted by the source goes through the lens. Find the power of the source needed to support the weight of the mirror.

(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a light wave is given by `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`. Find the stopping potential when this light is used in an experiment on photoelectric effect with the emitter having work function 1.9 eV.
The figure is the plot of stopping potential versus the frequency of the light used in an experiment on photoelectric effect. Find (a) the ratio h/e and (b) the work function.

Define the term: stopping potential in the photoelectric effect.
Do all the electrons that absorb a photon come out as photoelectrons?
If photons of ultraviolet light of energy 12 eV are incident on a metal surface of work function of 4 eV, then the stopping potential (in eV) will be :
How would the stopping potential for a given photosensitive surface change if the frequency of the incident radiation were increased? Justify your answer.
- Assertion (A): For the radiation of a frequency greater than the threshold frequency, the photoelectric current is proportional to the intensity of the radiation.
- Reason (R): Greater the number of energy quanta available, the greater the number of electrons absorbing the energy quanta and the greater the number of electrons coming out of the metal.
