Advertisements
Advertisements
प्रश्न
Monochromatic radiation of wavelength 640.2 nm (1 nm = 10−9 m) from a neon lamp irradiates photosensitive material made of caesium on tungsten. The stopping voltage is measured to be 0.54 V. The source is replaced by an iron source and its 427.2 nm line irradiates the same photo-cell. Predict the new stopping voltage.
Advertisements
उत्तर
Wavelength of the monochromatic radiation, λ = 640.2 nm = 640.2 × 10−9 m
Stopping potential of the neon lamp, V0 = 0.54 V
Charge on an electron, e = 1.6 × 10−19 C
Planck’s constant, h = 6.6 × 10−34 Js
Let `phi_0` be the work function and ν be the frequency of emitted light.
We have the photo-energy relation from the photoelectric effect as:
eV0 = hv − `phi_0`
`phi_0 = "hc"/lambda - "eV"_0`
= `(6.6 xx 10^(-34) xx 3 xx 10^8)/(640.2 xx 10^(-9)) - 1.6 xx 10^(-19) xx 0.54`
= `3.093 xx 10^(-19) - 0.864 xx 10^(-19)`
= `2.229 xx 10^(-19) "J"`
`= (2.229 xx 10^(-19))/(1.6 xx 10^(-19))`
= 1.39 eV
Wavelength of the radiation emitted from an iron source, λ' = 427.2 nm
= 427.2 × 10−9 m
Let `"V"_0^"'"` be the new stopping potential. Hence, photo-energy is given as:
`"eV"_0^"'" = "hc"/(lambda"'") - phi_0`
= `(6.6 xx 10^(-34) xx 3 xx 10^(8))/(427.2 xx 10^(-9)) - 2.229 xx 10^(-19)`
= `4.63 xx 10^(-19) - 2.229 xx 10^(-19)`
= `2.401 xx 10^(-19) "J"`
`= (2.401 xx 10^(-19))/(1.6 xx 10^(-19))`
= 1.5 eV
Hence, the new stopping potential is 1.50 eV.
APPEARS IN
संबंधित प्रश्न
The photoelectric cut-off voltage in a certain experiment is 1.5 V. What is the maximum kinetic energy of photoelectrons emitted?
Draw graphs showing variation of photoelectric current with applied voltage for two incident radiations of equal frequency and different intensities. Mark the graph for the radiation of higher intensity.
Is it always true that for two sources of equal intensity, the number of photons emitted in a given time are equal?
Let nr and nb be the number of photons emitted by a red bulb and a blue bulb, respectively, of equal power in a given time.
A point source causes photoelectric effect from a small metal plate. Which of the following curves may represent the saturation photocurrent as a function of the distance between the source and the metal?
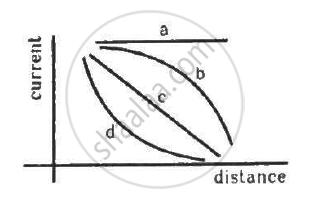
When the intensity of a light source in increased,
(a) the number of photons emitted by the source in unit time increases
(b) the total energy of the photons emitted per unit time increases
(c) more energetic photons are emitted
(d) faster photons are emitted
A beam of white light is incident normally on a plane surface absorbing 70% of the light and reflecting the rest. If the incident beam carries 10 W of power, find the force exerted by it on the surface.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, find the force exerted by the light beam on the sphere.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, Show that the force on the sphere due to the light falling on it is the same even if the sphere is not perfectly absorbing.
Find the maximum kinetic energy of the photoelectrons ejected when light of wavelength 350 nm is incident on a cesium surface. Work function of cesium = 1.9 eV
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a monochromatic beam is 1.2 × 1015 times per second. Find the maximum kinetic energy of the photoelectrons when this light falls on a metal surface whose work function is 2.0 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Define the term: threshold frequency
Answer the following question.
Plot a graph of photocurrent versus anode potential for radiation of frequency ν and intensities I1 and I2 (I1 < I2).
In photoelectric effect the photo current ______.
Consider a metal exposed to light of wavelength 600 nm. The maximum energy of the electron doubles when light of wavelength 400 nm is used. Find the work function in eV.
Two monochromatic beams A and B of equal intensity I, hit a screen. The number of photons hitting the screen by beam A is twice that by beam B. Then what inference can you make about their frequencies?
Consider a thin target (10–2 cm square, 10–3 m thickness) of sodium, which produces a photocurrent of 100 µA when a light of intensity 100W/m2 (λ = 660 nm) falls on it. Find the probability that a photoelectron is produced when a photons strikes a sodium atom. [Take density of Na = 0.97 kg/m3].
If photons of ultraviolet light of energy 12 eV are incident on a metal surface of work function of 4 eV, then the stopping potential (in eV) will be :
Read the following paragraph and answer the questions.
| The figure shows the variation of photoelectric current measured in a photocell circuit as a function of the potential difference between the plates of the photocell when light beams A, B, C and D of different wavelengths are incident on the photocell. Examine the given figure and answer the following questions: |

- Which light beam has the highest frequency and why?
- Which light beam has the longest wavelength and why?
- Which light beam ejects photoelectrons with maximum momentum and why?
