Advertisements
Advertisements
प्रश्न
Do all the electrons that absorb a photon come out as photoelectrons?
Advertisements
उत्तर
The photoelectric effect is the emission of electrons (called photo-electrons when light strikes a surface. To escape from the surface, the electron must absorb enough energy from the incident radiation to overcome the attraction of positive ions in the material of the surface.
The photoelectric effect is based on the principle of conservation of energy.
1. Two conducting electrodes, the anode (Q) and cathode (P) are enclosed in an evacuated glass tube as shown on next page.
2. The battery or other source of potential difference creates an electric field in the direction from anode to cathode.
3. Light of a certain wavelength or frequency falling on the surface of the cathode causes a current in the external circuit called photoelectric current.
4. As the potential difference increases, photoelectric current also increases till saturation is reached.
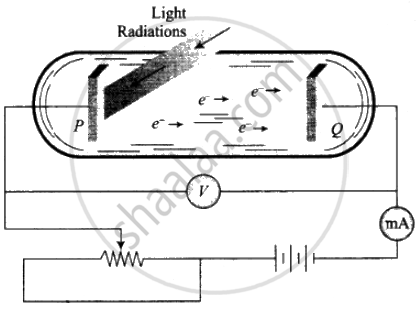
5. When polarity of the battery is reversed (i.e., plate Q is at negative potential w.r.t. plate P) electrons start moving back towards the cathode.
6. At a particular negative potential of plate Q, no electron will reach the plate Q and the current will become zero. This negative potential is called stopping potential denoted by V0. Maximum kinetic energy of photoelectrons in terms of stopping potential will therefore be Kmax = (|V0|) eV
So we conclude that in the photoelectric effect, we can observe that most electrons get scattered into the metal by absorbing a photon.
Therefore, all the electrons that absorb a photon don't come out as photoelectron. Only a few come out of metal whose energy becomes greater than the work function of the metal.
APPEARS IN
संबंधित प्रश्न
The photoelectric cut-off voltage in a certain experiment is 1.5 V. What is the maximum kinetic energy of photoelectrons emitted?
Can a photon be deflected by an electric field? Or by a magnetic field?
If an electron has a wavelength, does it also have a colour?
Let nr and nb be the number of photons emitted by a red bulb and a blue bulb, respectively, of equal power in a given time.
The equation E = pc is valid
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
If the frequency of light in a photoelectric experiment is doubled, the stopping potential will ______.
In which of the following situations, the heavier of the two particles has smaller de Broglie wavelength? The two particles
(a) move with the same speed
(b) move with the same linear momentum
(c) move with the same kinetic energy
(d) have fallen through the same height
Calculate the momentum of a photon of light of wavelength 500 nm.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The work function of a photoelectric material is 4.0 eV. (a) What is the threshold wavelength? (b) Find the wavelength of light for which the stopping potential is 2.5 V.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
In an experiment on photoelectric effect, the stopping potential is measured for monochromatic light beams corresponding to different wavelengths. The data collected are as follows:-
Wavelength (nm): 350 400 450 500 550
Stopping potential (V): 1.45 1.00 0.66 0.38 0.16
Plot the stopping potential against inverse of wavelength (1/λ) on a graph paper and find (a) Planck's constant (b) the work function of the emitter and (c) the threshold wavelength.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a monochromatic beam is 1.2 × 1015 times per second. Find the maximum kinetic energy of the photoelectrons when this light falls on a metal surface whose work function is 2.0 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A small piece of cesium metal (φ = 1.9 eV) is kept at a distance of 20 cm from a large metal plate with a charge density of 1.0 × 10−9 C m−2 on the surface facing the cesium piece. A monochromatic light of wavelength 400 nm is incident on the cesium piece. Find the minimum and maximum kinetic energy of the photoelectrons reaching the large metal plate. Neglect any change in electric field due to the small piece of cesium present.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
In the case of photoelectric effect experiment, explain the following facts, giving reasons.
The photoelectric current increases with increase of intensity of incident light.
Two monochromatic beams A and B of equal intensity I, hit a screen. The number of photons hitting the screen by beam A is twice that by beam B. Then what inference can you make about their frequencies?
The work function for a metal surface is 4.14 eV. The threshold wavelength for this metal surface is ______.
Why it is the frequency and not the intensity of the light source that determines whether the emission of photoelectrons will occur or not? Explain.
How would the stopping potential for a given photosensitive surface change if the frequency of the incident radiation were increased? Justify your answer.
A metallic plate exposed to white light emits electrons. For which of the following colours of light, the stopping potential will be maximum?
