Advertisements
Advertisements
प्रश्न
The work function of a photoelectric material is 4.0 eV. (a) What is the threshold wavelength? (b) Find the wavelength of light for which the stopping potential is 2.5 V.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Advertisements
उत्तर
Work function of a photoelectric material, ϕ = 4 eV = 4 × 1.6 × 10−19 J
Stopping potential, V0 = 2.5 V
Planck's constant, `h = 6.63 xx 10^-34 "Js"`
(a) Work function of a photoelectric material,
`phi = (hc)/λ_0`
Here, λ0 = threshold wavelength of light
c = speed of light
`therefore λ_0 = (hc)/phi`
`λ_0 = (6.63 xx 10^-34 xx 3 xx 10^8)/(4 xx 1.6 xx 10^-19)`
`λ_0 = (6.63 xx 3)/64 xx (10^27)/(10^-9)`
`λ_0 = 3.1 xx 10^-7 "m"`
`λ_0 = 310 "nm"`
(b) From Einstein's photoelectric equation,
`E = phi + eV_0`
On substituting the respective values , we get :-
`(hc)/λ = 4 xx 1.6 xx 10^-19 + 1.6 xx 10^-19 xx 2.5`
`⇒ λ = (6.63 xx 10^-34 xx 3 xx 10^8)/(6.5 xx 1.6 xx 10^-19)`
`⇒ λ = (6.63 xx 3 xx 10^-26)/(1.6 xx 10^-19 xx 6.5)`
`⇒ λ = 1.9125 xx 10^-7 = 191 "nm"`
APPEARS IN
संबंधित प्रश्न
Define the term 'intensity of radiation' in terms of photon picture of light.
The photoelectric cut-off voltage in a certain experiment is 1.5 V. What is the maximum kinetic energy of photoelectrons emitted?
A mercury lamp is a convenient source for studying frequency dependence of photoelectric emission, since it gives a number of spectral lines ranging from the UV to the red end of the visible spectrum. In our experiment with rubidium photo-cell, the following lines from a mercury source were used:
λ1 = 3650 Å, λ2 = 4047 Å, λ3 = 4358 Å, λ4 = 5461 Å, λ5 = 6907 Å,
The stopping voltages, respectively, were measured to be:
V01 = 1.28 V, V02 = 0.95 V, V03 = 0.74 V, V04 = 0.16 V, V05 = 0 V
Determine the value of Planck’s constant h, the threshold frequency and work function for the material.
[Note: You will notice that to get h from the data, you will need to know e (which you can take to be 1.6 × 10−19 C). Experiments of this kind on Na, Li, K, etc. were performed by Millikan, who, using his own value of e (from the oil-drop experiment) confirmed Einstein’s photoelectric equation and at the same time gave an independent estimate of the value of h.]
The work function for the following metals is given:
Na: 2.75 eV; K: 2.30 eV; Mo: 4.17 eV; Ni: 5.15 eV
Which of these metals will not give photoelectric emission for a radiation of wavelength 3300 Å from a He-Cd laser placed 1 m away from the photocell? What happens if the laser is brought nearer and placed 50 cm away?
The following graph shows the variation of photocurrent for a photosensitive metal :
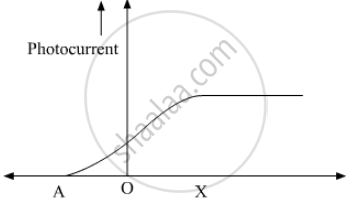
(a) Identify the variable X on the horizontal axis.
(b) What does the point A on the horizontal axis represent?
(c) Draw this graph for three different values of frequencies of incident radiation v1, v2 and v3 (v1 > v2 > v3) for same intensity.
(d) Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I1 > I2 > I3) having same frequency.
Draw graphs showing variation of photoelectric current with applied voltage for two incident radiations of equal frequency and different intensities. Mark the graph for the radiation of higher intensity.
Is it always true that for two sources of equal intensity, the number of photons emitted in a given time are equal?
What is the speed of a photon with respect to another photon if (a) the two photons are going in the same direction and (b) they are going in opposite directions?
Can a photon be deflected by an electric field? Or by a magnetic field?
Should the energy of a photon be called its kinetic energy or its internal energy?
Let nr and nb be the number of photons emitted by a red bulb and a blue bulb, respectively, of equal power in a given time.
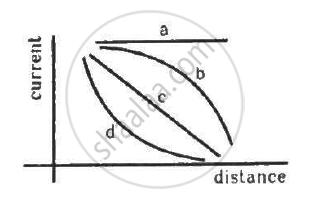
A point source causes photoelectric effect from a small metal plate. Which of the following curves may represent the saturation photocurrent as a function of the distance between the source and the metal?
Photoelectric effect supports quantum nature of light because
(a) there is a minimum frequency below which no photoelectrons are emitted
(b) the maximum kinetic energy of photoelectrons depends only on the frequency of light and not on its intensity
(c) even when the metal surface is faintly illuminated the photoelectrons leave the surface immediately
(d) electric charge of the photoelectrons is quantised
In which of the following situations, the heavier of the two particles has smaller de Broglie wavelength? The two particles
(a) move with the same speed
(b) move with the same linear momentum
(c) move with the same kinetic energy
(d) have fallen through the same height
The work function of a metal is 2.5 × 10−19 J. (a) Find the threshold frequency for photoelectric emission. (b) If the metal is exposed to a light beam of frequency 6.0 × 1014 Hz, what will be the stopping potential?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a light wave is given by `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`. Find the stopping potential when this light is used in an experiment on photoelectric effect with the emitter having work function 1.9 eV.
The electric field associated with a light wave is given by `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`. Find the stopping potential when this light is used in an experiment on photoelectric effect with the emitter having work function 1.9 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The graph shows the variation of photocurrent for a photosensitive metal
- What does X and A on the horizontal axis represent?
- Draw this graph for three different values of frequencies of incident radiation ʋ1, ʋ2 and ʋ3 (ʋ3 > ʋ2 > ʋ1) for the same intensity.
- Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I3 > I2 > I1) having the same frequency.
Read the following paragraph and answer the questions.
| The figure shows the variation of photoelectric current measured in a photocell circuit as a function of the potential difference between the plates of the photocell when light beams A, B, C and D of different wavelengths are incident on the photocell. Examine the given figure and answer the following questions: |

- Which light beam has the highest frequency and why?
- Which light beam has the longest wavelength and why?
- Which light beam ejects photoelectrons with maximum momentum and why?
