Advertisements
Advertisements
प्रश्न
In which reaction mechanism carbocation is formed?
विकल्प
SN1
SN2
Both SN1 and SN2
None of them
Advertisements
उत्तर
SN1
APPEARS IN
संबंधित प्रश्न
Out of  , which is more reactive towards SN1 reaction and why?
, which is more reactive towards SN1 reaction and why?
Which would undergo SN2 reaction faster in the following pair and why ?

Which compound in the following pair will react faster in SN2 reaction with OH−?
CH3Br or CH3I
How will you bring about the following conversion?
Toluene to benzyl alcohol
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
What happens when methyl chloride is treated with KCN?
C–Cl bond length in chlorobenzene is shorter than C–Cl bond length in CH3–Cl.
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Identify 'A' in the following reaction -

(a) 2- Bromo-2 methylbutane
(b) 1 -Bromo-2,2-dimethylpropane
(c) 1 - Bromo - 3 -methylbutane
(d) 1 - Bromo- 2 -methylpropane
What is the action of the following on ethyl bromide
alcoholic solution of potassium hydroxide.
What is the action of the following on ethyl bromide?
moist silver oxide
SN1 reaction of alkyl halides lead to ___________.
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
The increasing order of nucleophilicity would be:
The reaction of C6H5–CH=CH–CH3 with HBr produces:
Which of the following compounds will give a racemic mixture on nucleophilic substitution by OH ion?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Read the passage given below and answer the following question:
Nucleophilic substitution reaction of haloalkane can be conducted according to both SN1 and SN2 mechanisms. However, which mechanism it is based on is related to such factors as the structure of haloalkane, and properties of leaving group, nucleophilic reagent and solvent.
Influences of halogen: No matter which mechanism the nucleophilic substitution reaction is based on, the leaving group always leave the central carbon atom with electron pair. This is just the opposite of the situation that nucleophilic reagent attacks the central carbon atom with electron pair. Therefore, the weaker the alkalinity of leaving group is, the more stable the anion formed is and it will be more easier for the leaving group to leave the central carbon atom; that is to say, the reactant is more easier to be substituted. The alkalinity order of halogen ion is I− < Br− < Cl− < F− and the order of their leaving tendency should be I− > Br− > Cl− > F−. Therefore, in four halides with the same alkyl and different halogens, the order of substitution reaction rate is RI > RBr > RCl > RF. In addition, if the leaving group is very easy to leave, many carbocation intermediates are generated in the reaction and the reaction is based on SN1 mechanism. If the leaving group is not easy to leave, the reaction is based on SN2 a mechanism.
Influences of solvent polarity: In SN1 reaction, the polarity of the system increases from the reactant to the transition state, because polar solvent has a greater stabilizing effect on the transition state than the reactant, thereby reduce activation energy and accelerate the reaction. In SN2 reaction, the polarity of the system generally does not change from the reactant to the transition state and only charge dispersion occurs. At this time, polar solvent has a great stabilizing effect on Nu than the transition state, thereby increasing activation energy and slow down the reaction rate. For example, the decomposition rate (SN1) of tertiary chlorobutane in 25℃ water (dielectric constant 79) is 300000 times faster than in ethanol (dielectric constant 24). The reaction rate (SN2) of 2-bromopropane and NaOH in ethanol containing 40% water is twice slower than in absolute ethanol. In a word, the level of solvent polarity has influence on both SN1 and SN2 reactions, but with different results. Generally speaking, weak polar solvent is favorable for SN2 reaction, while strong polar solvent is favorable for SN1 reaction, because only under the action of polar solvent can halogenated hydrocarbon dissociate into carbocation and halogen ion and solvents with a strong polarity is favorable for solvation of carbocation, increasing its stability. Generally speaking, the substitution reaction of tertiary haloalkane is based on SN1 mechanism in solvents with a strong polarity (for example, ethanol containing water).
SN1 mechanism is favoured in which of the following solvents:
Read the passage given below and answer the following question:
Nucleophilic substitution reaction of haloalkane can be conducted according to both SN1 and SN2 mechanisms. However, which mechanism it is based on is related to such factors as the structure of haloalkane, and properties of leaving group, nucleophilic reagent and solvent.
Influences of halogen: No matter which mechanism the nucleophilic substitution reaction is based on, the leaving group always leave the central carbon atom with electron pair. This is just the opposite of the situation that nucleophilic reagent attacks the central carbon atom with electron pair. Therefore, the weaker the alkalinity of leaving group is, the more stable the anion formed is and it will be more easier for the leaving group to leave the central carbon atom; that is to say, the reactant is more easier to be substituted. The alkalinity order of halogen ion is I− < Br− < Cl− < F− and the order of their leaving tendency should be I− > Br− > Cl− > F−. Therefore, in four halides with the same alkyl and different halogens, the order of substitution reaction rate is RI > RBr > RCl > RF. In addition, if the leaving group is very easy to leave, many carbocation intermediates are generated in the reaction and the reaction is based on SN1 mechanism. If the leaving group is not easy to leave, the reaction is based on SN2 a mechanism.
Influences of solvent polarity: In SN1 reaction, the polarity of the system increases from the reactant to the transition state, because polar solvent has a greater stabilizing effect on the transition state than the reactant, thereby reduce activation energy and accelerate the reaction. In SN2 reaction, the polarity of the system generally does not change from the reactant to the transition state and only charge dispersion occurs. At this time, polar solvent has a great stabilizing effect on Nu than the transition state, thereby increasing activation energy and slow down the reaction rate. For example, the decomposition rate (SN1) of tertiary chlorobutane in 25℃ water (dielectric constant 79) is 300000 times faster than in ethanol (dielectric constant 24). The reaction rate (SN2) of 2-bromopropane and NaOH in ethanol containing 40% water is twice slower than in absolute ethanol. In a word, the level of solvent polarity has influence on both SN1 and SN2 reactions, but with different results. Generally speaking, weak polar solvent is favorable for SN2 reaction, while strong polar solvent is favorable for SN1 reaction, because only under the action of polar solvent can halogenated hydrocarbon dissociate into carbocation and halogen ion and solvents with a strong polarity is favorable for solvation of carbocation, increasing its stability. Generally speaking, the substitution reaction of tertiary haloalkane is based on SN1 mechanism in solvents with a strong polarity (for example, ethanol containing water).
SN1 reaction will be fastest in which of the following solvents?
Compound ‘A’ with molecular formula \[\ce{C4H9Br}\] is treated with aq. \[\ce{KOH}\] solution. The rate of this reaction depends upon the concentration of the compound ‘A’ only. When another optically active isomer ‘B’ of this compound was treated with aq. \[\ce{KOH}\] solution, the rate of reaction was found to be dependent on concentration of compound and \[\ce{KOH}\] both.
(i) Write down the structural formula of both compounds ‘A’ and ‘B’.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
Elimination reactions (especially β-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
The major product formed in the following reaction is:

Which of the following compounds will show retention in configuration on nucleophile substitution by OH− ion?
In SN1 reactions, the correct order of reactivity for the following compounds:
CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is ______.
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
|
Nucleophilic Substitution: Influences of solvent polarity: The reaction rate (SN2) of 2-bromopropane and NaOH in ethanol containing 40% water is twice slower than in absolute ethanol. Hence the level of solvent polarity has an influence on both SN1 and SN2 reactions but with different results. Generally speaking, a weak polar solvent is favourable for SN2 reaction, while a strong polar solvent is favourable for SN1. Generally speaking, the substitution reaction of tertiary haloalkane is based on SN1 mechanism in solvents with a strong polarity (for example ethanol containing water). |
Answer the following questions:
(a) Why racemisation occurs in SN1? (1)
(b) Why is ethanol less polar than water? (1)
(c) Which one of, the following in each pair is more reactive towards SN2 reaction? (2)
(i) CH3 – CH2 – I or CH3CH2 – Cl
(ii)
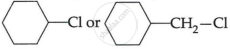
OR
(c) Arrange the following in the increasing order of their reactivity towards SN1 reactions: (2)
(i) 2-Bromo-2-methylbutane, 1-Bromo-pentane, 2-Bromo-pentane
(ii) 1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 2-Bromo-3- methylbutane
Acetic anhydride from acetic acid
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}\ce{CH3CH2CHCH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Br}\\
\end{array}\] or \[\begin{array}{cc}\phantom{.......}\ce{CH3}\\
\phantom{...}|\\
\ce{H3C - C - Br}\\
\phantom{...}|\\
\phantom{.......}\ce{CH3}\\
\end{array}\]
