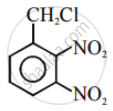
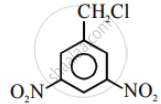
Advertisements
Advertisements
प्रश्न
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
Advertisements
उत्तर
In SN1 reaction, formation of carbocation as an intermediate takes place. This carbocation has sp2-hybridised and planar structure. This planar carbocation is attacked by nucleophile from both the sides equally to form d and l isomers in equal proportion. Such products are called racemic mixture. Hence, SN1 reactions are accompanied by racemisation in optically active alkyl halides.
संबंधित प्रश्न
What happens when methyl chloride is treated with KCN?
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Which compound in the following pair reacts faster in SN2 reaction with OH–?
- CH3Br or CH3
- CH3Cl, (CH3)3CCl
An organic molecule necessarily shows optical activity if it ____________.
Identify the end product (C) in the following sequence:
\[\ce{C2H5OH ->[SOCl2][Pyridine] A ->[KCN {(alc.)}] B ->[2H2O/H^+] C}\]
Identify X and Y in the following sequence:
\[\ce{C2H5Br ->[X] Product ->[Y] C3H7NH2}\]
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.
Retention of configuration is observed in ______.
Which alkyl halide from the following pair would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
\[\begin{array}{cc}
\ce{CH3CHCH2CH2Br}\\
|\phantom{.............}\\
\ce{CH3}\phantom{..........}\\
\end{array}\] or \[\begin{array}{cc}
\ce{CH3CH2CHCH2Br}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]