Advertisements
Advertisements
प्रश्न
Explain [Co(NH3)6]3+ is an inner orbital complex, whereas [Ni(NH3)6]2+ is an outer orbital complex.
[At. No.: Co = 27, Ni = 28]
Advertisements
उत्तर
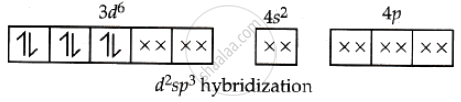
When a complex uses the inner (n − 1)d orbitals for hybridisation to form d2sp3 hybrid orbitals, it is known as an inner-orbital complex. The complex [Co(NH3)6]3+ uses inner (n − 1)d orbitals and is therefore classified as an inner orbital complex.
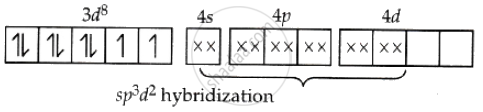
On the other hand, when a complex is formed using the outer d orbitals for hybridisation, resulting in 2sp3d2 hybrid orbitals, it is called an outer orbital complex. The complex [Ni(NH3)6]2+ uses the outer orbitals and is therefore an outer orbital complex.
[Co(NH3)6]3+ Co = 27 = 4s2 3d7
Ammonia, being a strong ligand, pairs.
[Ni(NH3)6]2+ Ni = 28 = 4s2 3d8
Although it is a strong ligand, if pairing of electrons occurs, only one d-orbital remains available, so the complex cannot undergo d2sp3 hybridisation.
संबंधित प्रश्न
Write the hybridization and shape of the following complexe : [CoF6]3–
(Atomic number : Co = 27, Ni = 28)
Write the hybridization and shape of the following complexe : [Ni(CN)4]2–
(Atomic number : Co = 27, Ni = 28)
[NiCl4]2− is paramagnetic while [Ni(CO)4] is diamagnetic though both are tetrahedral. Why?
[Fe(H2O)6]3+ is strongly paramagnetic whereas [Fe(CN)6]3− is weakly paramagnetic. Explain.
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
K[Cr(H2O)2(C2O4)2].3H2O
Amongst the following ions, which one has the highest magnetic moment value?
Write the hybridisation and magnetic character of [Co(C2O4)3]3–.
(At. no. of Co = 27)
Why is [NiCl4]2− paramagnetic while [Ni(CN)4]2− is diamagnetic? (Atomic number of Ni = 28)
Write the hybridization type and magnetic behaviour of the complex [Ni(CN)4]2−. (Atomic number of Ni = 28)
Write the hybridization and magnetic character of the following complexes:
[Fe(H2O)6]2+
(Atomic no. of Fe = 26)
Write the hybridization and magnetic character of the following complexes:
[Fe(CO)5]
(Atomic no. of Fe = 26)
Explain why \[\ce{[Fe(H2O)6]^{3+}}\] has magnetic moment value of 5.92 BM whereas \[\ce{[Fe(CN)6]^{3-}}\] – has a value of only 1.74 BM.
Why do compounds having similar geometry have different magnetic moment?
[Ti(H2O6]3+ T is paramagnetic is nature due to
The correct order of magnetic moment (spin only value in B.m.) is:
Explain [Fe(CN)6]3− is an inner orbital complex, whereas [FeF6]3− is an outer orbital complex.
[Atomic number: Fe = 26]
Given below are two statements:
Statement I: Ferromagnetism is considered as an extreme form of paramagnetism.
Statement II: The number of unpaired electrons in a Cr2+ ion (Z = 24) is the same as that of a Nd3+ ion (Z = 60).
In the light of the above statements, choose the correct answer from the options given below:
