Advertisements
Advertisements
प्रश्न
Explain [Fe(CN)6]3− is an inner orbital complex, whereas [FeF6]3− is an outer orbital complex.
[Atomic number: Fe = 26]
Advertisements
उत्तर
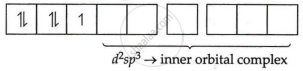
[Fe(CN)6]3−
Fe = 3 oxidation state
i.e., electronic configuration = 3d5
CNΘ = strong field ligand
i.e., Δ0 > P pairing will take place
Electronic configuration of Fe3+ = 3d5
[FeF6]3−
Fe = +3 oxidation state
F− = Weak field ligand Δ0 < P `->` Pairing will not take place.

APPEARS IN
संबंधित प्रश्न
For the complex [Fe(H2O)6]+3, write the hybridisation, magnetic character and spin of the complex. (At, number : Fe = 26)
Write the hybridization and shape of the following complexe : [Ni(CN)4]2–
(Atomic number : Co = 27, Ni = 28)
Write down the IUPAC name of the following complex and indicate the oxidation state, electronic configuration and coordination number. Also, give the stereochemistry and magnetic moment of the complex:
K[Cr(H2O)2(C2O4)2].3H2O
Write the hybridization and magnetic character of the following complexes:
[Fe(CO)5]
(Atomic no. of Fe = 26)
Magnetic moment of \[\ce{[MnCl4]^{2-}}\] is 5.92 BM. Explain giving reason.
Explain why \[\ce{[Fe(H2O)6]^{3+}}\] has magnetic moment value of 5.92 BM whereas \[\ce{[Fe(CN)6]^{3-}}\] – has a value of only 1.74 BM.
Assertion: \[\ce{[Fe(CN)6]^{3-}}\] ion shows magnetic moment corresponding to two unpaired electrons.
Reason: Because it has d2sp3 type hybridisation.
[Ti(H2O6]3+ T is paramagnetic is nature due to
The correct order of magnetic moment (spin only value in B.m.) is:
Given below are two statements:
Statement I: Ferromagnetism is considered as an extreme form of paramagnetism.
Statement II: The number of unpaired electrons in a Cr2+ ion (Z = 24) is the same as that of a Nd3+ ion (Z = 60).
In the light of the above statements, choose the correct answer from the options given below:
