Advertisements
Advertisements
प्रश्न
Explain [Fe(CN)6]3− is an inner orbital complex, whereas [FeF6]3− is an outer orbital complex.
[Atomic number: Fe = 26]
Advertisements
उत्तर
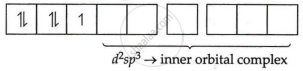
[Fe(CN)6]3−
Fe = 3 oxidation state
i.e., electronic configuration = 3d5
CNΘ = strong field ligand
i.e., Δ0 > P pairing will take place
Electronic configuration of Fe3+ = 3d5
[FeF6]3−
Fe = +3 oxidation state
F− = Weak field ligand Δ0 < P `->` Pairing will not take place.

APPEARS IN
संबंधित प्रश्न
For the complex [Fe(H2O)6]+3, write the hybridisation, magnetic character and spin of the complex. (At, number : Fe = 26)
Write the hybridization and shape of the following complexe : [CoF6]3–
(Atomic number : Co = 27, Ni = 28)
Write the hybridization and shape of the following complexe : [Ni(CN)4]2–
(Atomic number : Co = 27, Ni = 28)
[NiCl4]2− is paramagnetic while [Ni(CO)4] is diamagnetic though both are tetrahedral. Why?
Write the hybridisation and magnetic character of [Co(C2O4)3]3–.
(At. no. of Co = 27)
Write the hybridization type and magnetic behaviour of the complex [Ni(CN)4]2−. (Atomic number of Ni = 28)
Write the hybridization and magnetic character of the following complexes:
[Fe(CO)5]
(Atomic no. of Fe = 26)
Which of the following options are correct for \[\ce{[Fe(CN)6]^{3-}}\] complex?
(i) d2sp3 hybridisation
(ii) sp3d2 hybridisation
(iii) paramagnetic
(iv) diamagnetic
Why do compounds having similar geometry have different magnetic moment?
Assertion: \[\ce{[Fe(CN)6]^{3-}}\] ion shows magnetic moment corresponding to two unpaired electrons.
Reason: Because it has d2sp3 type hybridisation.
