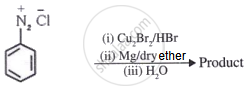Advertisements
Advertisements
प्रश्न
Illustrate Sandmeyer's reaction with an equation.
Advertisements
उत्तर
In benzene diazonium chloride (C6H5N2Cl) the Cl–, Br– and CN– nucleophiles can easily be introduced in the benzene ring in the presence of Cu(I) ion. This reaction is called the Sandmeyer reaction. It can be illustrated as

APPEARS IN
संबंधित प्रश्न
Write the structures of A, B and C in the following:

The reaction \[\ce{Ar\overset{+}{N2}Cl- ->[Cu/HCl] ArCl + N2 + CuCl}\] is anmed as ______.
Which of the following cannot be prepared by Sandmeyer’s reaction?
(i) Chlorobenzene
(ii) Bromobenzene
(iii) Iodobenzene
(iv) Fluorobenzene
Arrange the following compounds in increasing order of dipole moment.
\[\ce{CH3CH2CH3, CH3CH2NH2, CH3CH2OH}\]
Give reasons to support the answer:
3-Hydroxy pentan-2-one shows positive Tollen’s test.
The IUPAC name of the following compound is:
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\ce{OH}\phantom{.........}\\
|\phantom{......}|\phantom{...........}\\
\ce{CH3 - CH - CH - CH2 - COOH}\\
\end{array}\]
Which of the following statements are correct:
(P) C6H5N=CH-C6H5 is a Schiff’s base.
(Q) A dye is obtained by the reaction of aniline and C6H5N=NCl.
(R) C6H5CH2NH2 on treatment with [NaNO2 + HCl] gives diazonium salt.
(S) p-Toluidine on treatment with [HNO2 + HCl] gives diazonium salt.
In a set of reactions, nitrobenzene gave a product D. Identify the product D.

Write the structures of A, B and C in the following reaction:

Identify the product in the following reaction: