Advertisements
Advertisements
प्रश्न
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Sodium [At. no. = 11, Mass no. = 23]
Advertisements
उत्तर
Structure of atoms:
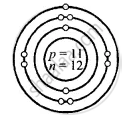
Z is Atomic Number and A is the mass number
Sodium \[\ce{^23_11Na}\]
Z = 11 = p = e
K L M
e = 11 = 2, 8, 1
A = p + n
23 = 11 + n
∴ n = 23 − 11 = 12
APPEARS IN
संबंधित प्रश्न
Fill in the blank of the following statement :
The total number of protons and neutrons in the nucleus of an atom is called its _________.
Fill in the following blanks in respect of an atom of an element
| No. of protons | No. of neutron | Mass number | Atomic number | No. of electrons | Symbol |
| 11 | 12 | ___________ | ___________ | ___________ | ________ |
What is the significance of atomic numbers in the modern Periodic Table?
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Oxygen – \[\ce{^16_8O}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Carbon [At. no. = 6, Mass no. = 12]
Define the term of atomic number.
From the symbol \[\ce{^4_2He}\] for the element helium, write down the mass number and the atomic number of the element.
Elements X, Y, Z have atomic numbers 6, 9 and 12 respectively. Which one:
- forms anion – negative ion;
- forms cation – positive ion;
- has 4 electrons in the outermost orbit. [Y, Z, X]
Complete the following table.
| Atomic Number | Mass Number | Number of Neutrons | Number of Protons | Number of Electrons | Name of the Element |
| 9 | ______ | 10 | ______ | ______ | ______ |
| 16 | ______ | 16 | ______ | ______ | ______ |
| ______ | 24 | ______ | ______ | 12 | Magnesium |
| ______ | 2 | ______ | 1 | ______ | ______ |
| ______ | 1 | 0 | 1 | 1 | ______ |
Match the following:
| Column A | Column B | ||
| 1. | Mass of proton | a. | +1 |
| 2. | Mass of electron | b. | -1 |
| 3. | Charge of electron | c. | 9.31 x 10-28g |
| 4. | Charge of proton | d. | 1.67 x 10-24g |
