Advertisements
Advertisements
प्रश्न
Draw the orbital diagram of `""_20^40"Ca"^(2+)` ion and state the number of three fundamental particles present in it.
Advertisements
उत्तर
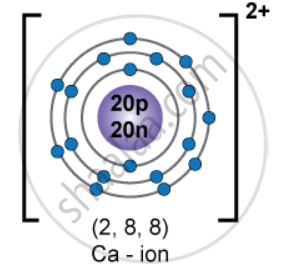
Neutral calcium (Z = 20) has
1s22s22p63s23p64s2
For Ca2+, remove 2 electrons from the outermost 4s orbital:
1s22s22p63s23p6
it has 18 electrons
-
Protons = 20
-
Neutrons = 20
-
Electrons = 18
APPEARS IN
संबंधित प्रश्न
Multiple Choice Questions
The sum of number of protons and number of neutrons present in the nucleus of an atom is called its
What is variable valency? Name two elements having variable valency and state their valencies.
The number of electrons in the atom of an element X is 15 and the number of neutrons is 16. Which of the following is the correct representation of an atom of this element ?
From the symbol `""_15^31"P"`, state : atomic number of phosphorus
The atom of an element is made up of 4 protons, 5 neutrons and 4 electrons. What are its atomic number and mass number?
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
If an isotope of ‘H’ has mass no. = 2, how many electrons does it have.
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
Which sub-atomic particles in the 3 isotopes of ‘H’ are the same.
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Lithium [At. no. = 3, Mass no. = 7]
What is mass number of an atom?
In a neutral atom number of protons is equal to the number of neutrons.
