Advertisements
Advertisements
Question
Draw the orbital diagram of `""_20^40"Ca"^(2+)` ion and state the number of three fundamental particles present in it.
Advertisements
Solution
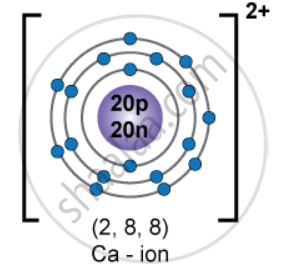
Neutral calcium (Z = 20) has
1s22s22p63s23p64s2
For Ca2+, remove 2 electrons from the outermost 4s orbital:
1s22s22p63s23p6
it has 18 electrons
-
Protons = 20
-
Neutrons = 20
-
Electrons = 18
APPEARS IN
RELATED QUESTIONS
Dalton said that ______ could not be divided.
FILL IN THE BLANK
Atomic theory of matter was given by ..........................
Name the particles which actually determine the mass of an atom.
Fill in the Blank of the Following Statement :
An atom has atomic mass number 23 and atomic number 11. The atom has .................... electrons.
Match the atomic numbers 4,8,10,15 and 19 with each of the following:
An element which does not form ion
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Argon – \[\ce{^40_18Ar}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
Which of the following are true for an element?
- Atomic number = number of protons + number of electrons
- Mass number = number of protons + number of neutrons
- Atomic mass = number of protons = number of neutrons
- Atomic number = number of protons = number of electrons
What is mass number of an atom?
Name the 3 sub-atomic particles of an atom.
______ = No. of protons = No. of the electrons.
