Advertisements
Advertisements
प्रश्न
Draw the orbital diagram of `""_20^40"Ca"^(2+)` ion and state the number of three fundamental particles present in it.
Advertisements
उत्तर
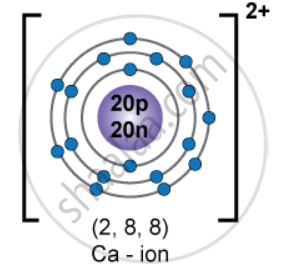
Neutral calcium (Z = 20) has
1s22s22p63s23p64s2
For Ca2+, remove 2 electrons from the outermost 4s orbital:
1s22s22p63s23p6
it has 18 electrons
-
Protons = 20
-
Neutrons = 20
-
Electrons = 18
APPEARS IN
संबंधित प्रश्न
With the help of given Table, find out the mass number of sulphur atom.
| Composition of Atoms of the First Eighteen Elements with Electron Distribution in Various Shells | ||||||||||
| Name of Element | Symbol | Atomic Number | Number of protons | Number of Neutrons | Number of Electrons | Distribution of Electrons | Valency | |||
| K | L | M | N | |||||||
| Hydrogen | H | 1 | 1 | - | 1 | 1 | - | - | - | 1 |
| Helium | He | 2 | 2 | 2 | 2 | 2 | - | - | - | 0 |
| Lithium | Li | 3 | 3 | 4 | 3 | 2 | 1 | - | - | 1 |
| Beryllium | Be | 4 | 4 | 5 | 4 | 2 | 2 | - | - | 2 |
| Boron | B | 5 | 5 | 6 | 5 | 2 | 3 | - | - | 3 |
| Carbon | C | 6 | 6 | 6 | 6 | 2 | 4 | - | - | 4 |
| Nitrogen | N | 7 | 7 | 7 | 7 | 2 | 5 | - | - | 3 |
| Oxygen | O | 8 | 8 | 8 | 8 | 2 | 6 | - | - | 2 |
| Fluorine | F | 9 | 9 | 10 | 9 | 2 | 7 | - | - | 1 |
| Neon | Ne | 10 | 10 | 10 | 10 | 2 | 8 | - | - | 0 |
| Sodium | Na | 11 | 11 | 12 | 11 | 2 | 8 | 1 | - | 1 |
| Magnesium | Mg | 12 | 12 | 12 | 12 | 2 | 8 | 2 | - | 2 |
| Aluminium | Al | 13 | 13 | 14 | 13 | 2 | 8 | 3 | - | 3 |
| Silicon | Si | 14 | 14 | 14 | 14 | 2 | 8 | 4 | - | 4 |
| Phosphorus | P | 15 | 15 | 16 | 15 | 2 | 8 | 5 | - | 3.5 |
| Sulphur | S | 16 | 16 | 16 | 16 | 2 | 8 | 6 | - | 2 |
| Chlorine | Cl | 17 | 17 | 18 | 17 | 2 | 8 | 7 | - | 1 |
| Argon | Ar | 18 | 18 | 22 | 18 | 2 | 8 | 8 | 0 | |
TRUE \ FALSE
After the emission of a beta-particle, the atomic number of the atom increases by one.
Fill in the blanks- The number of protons in the nucleus of an atom is called its ....................
The number of electrons in the atom of an element X is 15 and the number of neutrons is 16. Which of the following is the correct representation of an atom of this element ?
Match the atomic numbers 4,8,10,15 and 19 with each of the following:
An element which does not form ion
Name or state the following:
Elements having same mass number but different atomic number
State the main postulates of Dalton’s atomic theory.
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
Do isotopes have the same atomic number or the same mass number?
What are nucleons? How many nucleons are present in Phosphorous? Draw its structure.
What is the relation between mass number & atomic number?
