Advertisements
Advertisements
प्रश्न
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Isotopes of hydrogen [11H, 21H, 31H]
Advertisements
उत्तर
Structure of atoms:

\[\ce{^2_1H}\]

\[\ce{^3_1H}\]
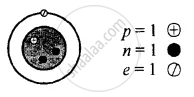
Z is Atomic Number and A is the mass number
Isotopes of hydrogen \[\ce{^1_1H}\]
Z = 1 = p = e
∴ e = 1
A = p + n
1 = 1 + n
∴ n = 1 − 1 = 0
APPEARS IN
संबंधित प्रश्न
Give the following a suitable word/phrase.
Atoms of same element with same atomic number but a different mass number.
FILL IN THE BLANK
Rutherford won the Nobel Prize in 1908 for his investigations in the field of ......................
The mass number of an element is 23 and it contains 11 electrons. What is the number of protons and neutrons in it? What is the atomic number of the element?
What is the significance of atomic numbers in the modern Periodic Table?
Elements X, Y and Z have atomic numbers 6,9 and 12 respectively. Which one:
- Forms an anion
- Forms a cation
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Argon – \[\ce{^40_18Ar}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
The atomic number and the mass number of an element is 26 and 56 respectively. Calculate the number of electrons, protons and neutrons in its atom. Draw the structure.
Show diagrammatically the electron distributions in a sodium atom and a sodium ion and also give their atomic number.
The particles that make up the atom are called ______ particles.
Match the following:
| Column A | Column B | ||
| 1. | K shell | a. | 8 |
| 2. | L shell | b. | 32 |
| 3. | M shell | c. | 2 |
| 4. | N shell | d. | 18 |
