Advertisements
Advertisements
Question
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Isotopes of hydrogen [11H, 21H, 31H]
Advertisements
Solution
Structure of atoms:

\[\ce{^2_1H}\]

\[\ce{^3_1H}\]
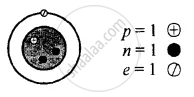
Z is Atomic Number and A is the mass number
Isotopes of hydrogen \[\ce{^1_1H}\]
Z = 1 = p = e
∴ e = 1
A = p + n
1 = 1 + n
∴ n = 1 − 1 = 0
APPEARS IN
RELATED QUESTIONS
With the help of given Table, find out the mass number of sulphur atom.
| Composition of Atoms of the First Eighteen Elements with Electron Distribution in Various Shells | ||||||||||
| Name of Element | Symbol | Atomic Number | Number of protons | Number of Neutrons | Number of Electrons | Distribution of Electrons | Valency | |||
| K | L | M | N | |||||||
| Hydrogen | H | 1 | 1 | - | 1 | 1 | - | - | - | 1 |
| Helium | He | 2 | 2 | 2 | 2 | 2 | - | - | - | 0 |
| Lithium | Li | 3 | 3 | 4 | 3 | 2 | 1 | - | - | 1 |
| Beryllium | Be | 4 | 4 | 5 | 4 | 2 | 2 | - | - | 2 |
| Boron | B | 5 | 5 | 6 | 5 | 2 | 3 | - | - | 3 |
| Carbon | C | 6 | 6 | 6 | 6 | 2 | 4 | - | - | 4 |
| Nitrogen | N | 7 | 7 | 7 | 7 | 2 | 5 | - | - | 3 |
| Oxygen | O | 8 | 8 | 8 | 8 | 2 | 6 | - | - | 2 |
| Fluorine | F | 9 | 9 | 10 | 9 | 2 | 7 | - | - | 1 |
| Neon | Ne | 10 | 10 | 10 | 10 | 2 | 8 | - | - | 0 |
| Sodium | Na | 11 | 11 | 12 | 11 | 2 | 8 | 1 | - | 1 |
| Magnesium | Mg | 12 | 12 | 12 | 12 | 2 | 8 | 2 | - | 2 |
| Aluminium | Al | 13 | 13 | 14 | 13 | 2 | 8 | 3 | - | 3 |
| Silicon | Si | 14 | 14 | 14 | 14 | 2 | 8 | 4 | - | 4 |
| Phosphorus | P | 15 | 15 | 16 | 15 | 2 | 8 | 5 | - | 3.5 |
| Sulphur | S | 16 | 16 | 16 | 16 | 2 | 8 | 6 | - | 2 |
| Chlorine | Cl | 17 | 17 | 18 | 17 | 2 | 8 | 7 | - | 1 |
| Argon | Ar | 18 | 18 | 22 | 18 | 2 | 8 | 8 | 0 | |
Fill in the blank
An ion which has a positive charge is called a ..................
Multiple Choice Questions
The sum of number of protons and number of neutrons present in the nucleus of an atom is called its
The atom of an element X has 7 electrons in its M shell.
What is the atomic number of element X?
Match the atomic numbers 4,8,10,15 and 19 with the following:
Element with 6 valence electrons
State why the atomic weight of an element is also termed relative atomic mass.
From the symbol \[\ce{^4_2He}\] for the element helium, write down the mass number and the atomic number of the element.
Complete the following table.
| Atomic Number | Mass Number | Number of Neutrons | Number of Protons | Number of Electrons | Name of the Element |
| 9 | ______ | 10 | ______ | ______ | ______ |
| 16 | ______ | 16 | ______ | ______ | ______ |
| ______ | 24 | ______ | ______ | 12 | Magnesium |
| ______ | 2 | ______ | 1 | ______ | ______ |
| ______ | 1 | 0 | 1 | 1 | ______ |
Show diagrammatically the electron distributions in a sodium atom and a sodium ion and also give their atomic number.
In which one of the following, the number of protons is greater than the number of neutrons but number of protons is less than the number of electrons?
