Advertisements
Advertisements
Question
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Sodium [At. no. = 11, Mass no. = 23]
Advertisements
Solution
Structure of atoms:
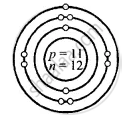
Z is Atomic Number and A is the mass number
Sodium \[\ce{^23_11Na}\]
Z = 11 = p = e
K L M
e = 11 = 2, 8, 1
A = p + n
23 = 11 + n
∴ n = 23 − 11 = 12
APPEARS IN
RELATED QUESTIONS
What are the atomic numbers of elements whose outermost electrons is represented by 2p3.
Give the following a suitable word/phrase.
Atoms of same element with same atomic number but a different mass number.
The atomic number and the mass number of sodium are 11 and 23 respectively. What information is conveyed by this statement.
FILL IN THE BLANK
Atomic theory of matter was given by ..........................
FILL IN THE BLANK
Rutherford won the Nobel Prize in 1908 for his investigations in the field of ......................
Give reason
Actual atomic mass is greater than mass number.
Match the atomic numbers 4,8,10,15 and 19 with the following:
Element which can form trivalent ion
Define the term of atomic number.
Show diagrammatically the electron distributions in a sodium atom and a sodium ion and also give their atomic number.
An element has 2 electrons in its N shell.
What is its atomic number?
