Advertisements
Advertisements
प्रश्न
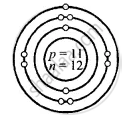
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Sodium [At. no. = 11, Mass no. = 23]
Advertisements
उत्तर
Structure of atoms:
Z is Atomic Number and A is the mass number
Sodium \[\ce{^23_11Na}\]
Z = 11 = p = e
K L M
e = 11 = 2, 8, 1
A = p + n
23 = 11 + n
∴ n = 23 − 11 = 12
APPEARS IN
संबंधित प्रश्न
With the help of given Table, find out the mass number of oxygen.
| Composition of Atoms of the First Eighteen Elements with Electron Distribution in Various Shells | ||||||||||
| Name of Element | Symbol | Atomic Number | Number of protons | Number of Neutrons | Number of Electrons | Distribution of Electrons | Valency | |||
| K | L | M | N | |||||||
| Hydrogen | H | 1 | 1 | - | 1 | 1 | - | - | - | 1 |
| Helium | He | 2 | 2 | 2 | 2 | 2 | - | - | - | 0 |
| Lithium | Li | 3 | 3 | 4 | 3 | 2 | 1 | - | - | 1 |
| Beryllium | Be | 4 | 4 | 5 | 4 | 2 | 2 | - | - | 2 |
| Boron | B | 5 | 5 | 6 | 5 | 2 | 3 | - | - | 3 |
| Carbon | C | 6 | 6 | 6 | 6 | 2 | 4 | - | - | 4 |
| Nitrogen | N | 7 | 7 | 7 | 7 | 2 | 5 | - | - | 3 |
| Oxygen | O | 8 | 8 | 8 | 8 | 2 | 6 | - | - | 2 |
| Fluorine | F | 9 | 9 | 10 | 9 | 2 | 7 | - | - | 1 |
| Neon | Ne | 10 | 10 | 10 | 10 | 2 | 8 | - | - | 0 |
| Sodium | Na | 11 | 11 | 12 | 11 | 2 | 8 | 1 | - | 1 |
| Magnesium | Mg | 12 | 12 | 12 | 12 | 2 | 8 | 2 | - | 2 |
| Aluminium | Al | 13 | 13 | 14 | 13 | 2 | 8 | 3 | - | 3 |
| Silicon | Si | 14 | 14 | 14 | 14 | 2 | 8 | 4 | - | 4 |
| Phosphorus | P | 15 | 15 | 16 | 15 | 2 | 8 | 5 | - | 3.5 |
| Sulphur | S | 16 | 16 | 16 | 16 | 2 | 8 | 6 | - | 2 |
| Chlorine | Cl | 17 | 17 | 18 | 17 | 2 | 8 | 7 | - | 1 |
| Argon | Ar | 18 | 18 | 22 | 18 | 2 | 8 | 8 | 0 | |
The composition of the nuclei of two atomic species X and Y are given as under
| X | Y | |
| Protons | 6 | 6 |
| Neutrons | 6 | 8 |
Give the mass numbers of X and Y. What is the relation between the two species?
In Milikan’s experiment, static electric charge on the oil drops has been obtained by shining X-rays. If the static electric charge on the oil drop is - 1.282 × 10-18C, calculate the number of electrons present on it.
Write true or false for the following statement
An atom on the whole has a positive charge.
Tick the most appropriate answer.
Which of these have similar chemical properties but different set of physical properties and mass number?
- isobars
- elements
- isotopes
- none of these
From the symbol `""_15^31"P"`, state : atomic number of phosphorus
The properties of the elements are a periodic function of their __________.
State why the atomic weight of an element is also termed relative atomic mass.
Differentiate mass number from atomic number.
Name the 3 sub-atomic particles of an atom.
