Advertisements
Advertisements
प्रश्न
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Isotopes of hydrogen [11H, 21H, 31H]
Advertisements
उत्तर
Structure of atoms:

\[\ce{^2_1H}\]

\[\ce{^3_1H}\]
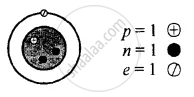
Z is Atomic Number and A is the mass number
Isotopes of hydrogen \[\ce{^1_1H}\]
Z = 1 = p = e
∴ e = 1
A = p + n
1 = 1 + n
∴ n = 1 − 1 = 0
APPEARS IN
संबंधित प्रश्न
Dalton said that ______ could not be divided.
FILL IN THE BLANK
Atomic theory of matter was given by ..........................
Fill in the blanks- The number of protons in the nucleus of an atom is called its ....................
Name or state the following:
Elements having same mass number but different atomic number
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Calcium – \[\ce{^40_20Ca}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
Which sub-atomic particles in the 3 isotopes of ‘H’ are the same.
Define the term of atomic number.
Electrons revolve around the nucleus in definite open shells.
Match the following:
| Column A | Column B | ||
| 1. | Mass of proton | a. | +1 |
| 2. | Mass of electron | b. | -1 |
| 3. | Charge of electron | c. | 9.31 x 10-28g |
| 4. | Charge of proton | d. | 1.67 x 10-24g |
What is the relation between mass number & atomic number?
