Advertisements
Advertisements
प्रश्न
State the mass number, the atomic number, number of neutrons and electronic configuration of the following atoms.
`""^12""_6C, ""^16""_8O, ""^19""_9F, ""^20""_10Ne, ""_13""Al""^27, ""_17""Cl""^35`
Also, draw atomic diagrams for them.
Advertisements
उत्तर
| Name of elements | Atomic number | Atomic mass |
No of proton |
No of electrons |
no.of neutrons |
Electronic configuration |
| `""^12""_6C` | 6 | 12 | 6 | 6 | 6 | 2, 4 |
| `""^16""_8O` | 8 | 16 | 8 | 8 | 8 | 2, 6 |
| `""^19""_9F` | 9 | 19 | 9 | 9 | 10 | 2, 7 |
| `""^20""_10Ne` | 10 | 20 | 10 | 10 | 10 | 2, 8 |
| `""_13""Al""^27` | 13 | 27 | 13 | 13 | 14 | 2, 8, 3 |
| `""_17""Cl""^35` | 17 | 35 | 17 | 17 | 18 | 2, 8, 7 |
| Carbon `""^12""_6C` | 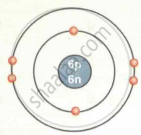 |
| Oxygen `""^16""_8O` |  |
| Fluorine `""^19""_9F` | 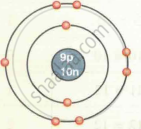 |
| Neon `""^20""_10Ne` |  |
| Aluminium `""_13""Al""^27` | 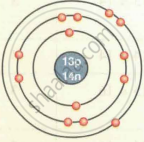 |
| Chlorine `""_17""Cl""^35` |  |
APPEARS IN
संबंधित प्रश्न
Complete the following table.
| Atomic number | Mass number | Number of Neutrons | Number of protons | Number of electrons | Name of the Atomic species |
| 9 | - | 10 | - | - | - |
| 16 | 32 | - | - | - | Sulphur |
| - | 24 | - | 12 | - | - |
| - | 2 | - | 1 | - | - |
| - | 1 | 0 | 1 | 0 | - |
What is variable valency? Name two elements having variable valency and state their valencies.
The mass number of an element is 18. It contains 7 electrons. What is the number of protons and neutrons in it? What is the atomic number of the element?
(a) What is meant by (i) atomic number, and (ii) mass number, of an element? Explain with the help of an example.
(b) What is the relation between the atomic number and mass number of an element?
(c) If an element M has mass number 24 and atomic number 12, how many neutrons does its atom contain?
From the symbol `""_15^31"P"`, state :
(i) mass number of phosphorus,
(ii) atomic number of phosphorus, and
(iii) electron configuration of phosphorus.
Draw the orbital diagram of `""_20^40"Ca"^(2+)` ion and state the number of three fundamental particles present in it.
If an atom ‘A’ has atomic number 19 and mass number 39, state –
- Its electronic configuration.
- The number of valence electrons it possesses.
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
If an isotope of ‘H’ has mass no. = 3, how many neutrons does it have.
K and L shells can accommodate a maximum of 2 and 8 electrons respectively.
What is the relation between mass number & atomic number?
