Advertisements
Advertisements
प्रश्न
State the mass number, the atomic number, number of neutrons and electronic configuration of the following atoms.
`""^12""_6C, ""^16""_8O, ""^19""_9F, ""^20""_10Ne, ""_13""Al""^27, ""_17""Cl""^35`
Also, draw atomic diagrams for them.
Advertisements
उत्तर
| Name of elements | Atomic number | Atomic mass |
No of proton |
No of electrons |
no.of neutrons |
Electronic configuration |
| `""^12""_6C` | 6 | 12 | 6 | 6 | 6 | 2, 4 |
| `""^16""_8O` | 8 | 16 | 8 | 8 | 8 | 2, 6 |
| `""^19""_9F` | 9 | 19 | 9 | 9 | 10 | 2, 7 |
| `""^20""_10Ne` | 10 | 20 | 10 | 10 | 10 | 2, 8 |
| `""_13""Al""^27` | 13 | 27 | 13 | 13 | 14 | 2, 8, 3 |
| `""_17""Cl""^35` | 17 | 35 | 17 | 17 | 18 | 2, 8, 7 |
| Carbon `""^12""_6C` | 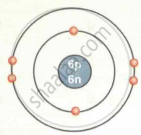 |
| Oxygen `""^16""_8O` |  |
| Fluorine `""^19""_9F` | 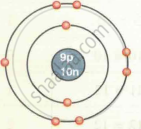 |
| Neon `""^20""_10Ne` |  |
| Aluminium `""_13""Al""^27` | 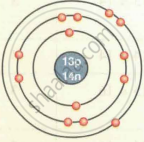 |
| Chlorine `""_17""Cl""^35` |  |
APPEARS IN
संबंधित प्रश्न
Explain the Mass number with example.
What are the atomic numbers of elements whose outermost electrons is represented by 3p5?
TRUE \ FALSE
After the emission of a beta-particle, the atomic number of the atom increases by one.
The mass number of an element is 23 and it contains 11 electrons. What is the number of protons and neutrons in it? What is the atomic number of the element?
The atomic number and mass number of sodium are 11 and 23 respectively. What information is conveyed by this statement?
Match the atomic numbers 4,8,10,15 and 19 with the following:
Element with four shells
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Calcium – \[\ce{^40_20Ca}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
If an isotope of ‘H’ has mass no. = 2, how many electrons does it have.
Draw the structure of the following atoms showing the nucleus containing – protons, neutrons and the orbits with the respective electron:
Sodium [At. no. = 11, Mass no. = 23]
Give short answer:
what is the atomic mass number? mention its formula.
