Advertisements
Advertisements
Question
State the mass number, the atomic number, number of neutrons and electronic configuration of the following atoms.
`""^12""_6C, ""^16""_8O, ""^19""_9F, ""^20""_10Ne, ""_13""Al""^27, ""_17""Cl""^35`
Also, draw atomic diagrams for them.
Advertisements
Solution
| Name of elements | Atomic number | Atomic mass |
No of proton |
No of electrons |
no.of neutrons |
Electronic configuration |
| `""^12""_6C` | 6 | 12 | 6 | 6 | 6 | 2, 4 |
| `""^16""_8O` | 8 | 16 | 8 | 8 | 8 | 2, 6 |
| `""^19""_9F` | 9 | 19 | 9 | 9 | 10 | 2, 7 |
| `""^20""_10Ne` | 10 | 20 | 10 | 10 | 10 | 2, 8 |
| `""_13""Al""^27` | 13 | 27 | 13 | 13 | 14 | 2, 8, 3 |
| `""_17""Cl""^35` | 17 | 35 | 17 | 17 | 18 | 2, 8, 7 |
| Carbon `""^12""_6C` | 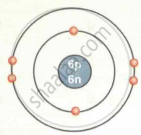 |
| Oxygen `""^16""_8O` |  |
| Fluorine `""^19""_9F` | 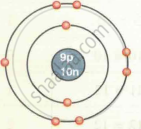 |
| Neon `""^20""_10Ne` |  |
| Aluminium `""_13""Al""^27` | 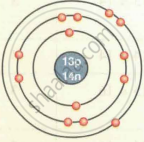 |
| Chlorine `""_17""Cl""^35` |  |
APPEARS IN
RELATED QUESTIONS
Fill in the blanks.
Isotopes are the atoms of ______ element having the ______ atomic number but ______ mass number.
Write true or false for the following statement
An atom on the whole has a positive charge.
Name the particles which actually determine the mass of an atom.
Fill in the blanks- The number of protons in the nucleus of an atom is called its ....................
The properties of the elements are a periodic function of their __________.
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Oxygen – \[\ce{^16_8O}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
What is atomic number?
What is a neutron?
A ______ number describes a specific aspect of an electron.
Mass No =______ + No. of Neutrons.
