Advertisements
Advertisements
Question
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Oxygen – \[\ce{^16_8O}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
Advertisements
Solution
Oxygen – \[\ce{^16_8O}\]
Atomic number Z = p = e = 8
Mass number A = p + n
16 = 8 + n
∴ n = 16 − 8 = 8
O
8 = 2, 6
(K, L)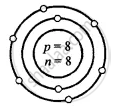
APPEARS IN
RELATED QUESTIONS
With the help of given Table, find out the mass number of oxygen.
| Composition of Atoms of the First Eighteen Elements with Electron Distribution in Various Shells | ||||||||||
| Name of Element | Symbol | Atomic Number | Number of protons | Number of Neutrons | Number of Electrons | Distribution of Electrons | Valency | |||
| K | L | M | N | |||||||
| Hydrogen | H | 1 | 1 | - | 1 | 1 | - | - | - | 1 |
| Helium | He | 2 | 2 | 2 | 2 | 2 | - | - | - | 0 |
| Lithium | Li | 3 | 3 | 4 | 3 | 2 | 1 | - | - | 1 |
| Beryllium | Be | 4 | 4 | 5 | 4 | 2 | 2 | - | - | 2 |
| Boron | B | 5 | 5 | 6 | 5 | 2 | 3 | - | - | 3 |
| Carbon | C | 6 | 6 | 6 | 6 | 2 | 4 | - | - | 4 |
| Nitrogen | N | 7 | 7 | 7 | 7 | 2 | 5 | - | - | 3 |
| Oxygen | O | 8 | 8 | 8 | 8 | 2 | 6 | - | - | 2 |
| Fluorine | F | 9 | 9 | 10 | 9 | 2 | 7 | - | - | 1 |
| Neon | Ne | 10 | 10 | 10 | 10 | 2 | 8 | - | - | 0 |
| Sodium | Na | 11 | 11 | 12 | 11 | 2 | 8 | 1 | - | 1 |
| Magnesium | Mg | 12 | 12 | 12 | 12 | 2 | 8 | 2 | - | 2 |
| Aluminium | Al | 13 | 13 | 14 | 13 | 2 | 8 | 3 | - | 3 |
| Silicon | Si | 14 | 14 | 14 | 14 | 2 | 8 | 4 | - | 4 |
| Phosphorus | P | 15 | 15 | 16 | 15 | 2 | 8 | 5 | - | 3.5 |
| Sulphur | S | 16 | 16 | 16 | 16 | 2 | 8 | 6 | - | 2 |
| Chlorine | Cl | 17 | 17 | 18 | 17 | 2 | 8 | 7 | - | 1 |
| Argon | Ar | 18 | 18 | 22 | 18 | 2 | 8 | 8 | 0 | |
(a) What is meant by (i) atomic number, and (ii) mass number, of an element? Explain with the help of an example.
(b) What is the relation between the atomic number and mass number of an element?
(c) If an element M has mass number 24 and atomic number 12, how many neutrons does its atom contain?
The atom of an element is made up of 4 protons, 5 neutrons and 4 electrons. What are its atomic number and mass number?
From the symbol `""_2^4"He"` for the element helium, write down the mass number and the atomic number of the element.
From the symbol \[\ce{^4_2He}\] for the element helium, write down the mass number and the atomic number of the element.
What are nucleons? How many nucleons are present in Phosphorous? Draw its structure.
What is mass number of an atom?
Mass No. 27 is for the element ______.
In a neutral atom number of protons is equal to the number of neutrons.
If M is the atomic mass and A is the mass number, then packing fraction is given by ______.
