Advertisements
Advertisements
प्रश्न
Draw the atomic diagram of the following element showing the distribution of – protons, neutrons and the electrons in the various shell of the atom.
Oxygen – \[\ce{^16_8O}\]
[The upper number represent the – mass number and the lower number represent the – atomic number e.g. calcium – mass number = 40, atomic number = 20]
Advertisements
उत्तर
Oxygen – \[\ce{^16_8O}\]
Atomic number Z = p = e = 8
Mass number A = p + n
16 = 8 + n
∴ n = 16 − 8 = 8
O
8 = 2, 6
(K, L)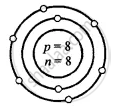
APPEARS IN
संबंधित प्रश्न
Complete the following table.
| Atomic number | Mass number | Number of Neutrons | Number of protons | Number of electrons | Name of the Atomic species |
| 9 | - | 10 | - | - | - |
| 16 | 32 | - | - | - | Sulphur |
| - | 24 | - | 12 | - | - |
| - | 2 | - | 1 | - | - |
| - | 1 | 0 | 1 | 0 | - |
Which of the following are isoelectronic species i.e., those having the same number of electrons?
Na+, K+, Mg2+, Ca2+, S2–, Ar
Fill in the blanks.
Isotopes are the atoms of ______ element having the ______ atomic number but ______ mass number.
What are the two main features of Rutherford’s atomic model?
Tick the most appropriate answer.
Which of these have similar chemical properties but different set of physical properties and mass number?
- isobars
- elements
- isotopes
- none of these
An element has an atomic number 12. How many electrons will be present in the K, L and M energy shells of its atom?
Draw the orbital diagram of `""_20^40"Ca"^(2+)` ion and state the number of three fundamental particles present in it.
The atomic number of an element is 9, it has 10 neutrons. Find the element from the periodic table. What will be its mass number?
What are nucleons? How many nucleons are present in Phosphorous? Draw its structure.
What is mass number of an atom?
