Please select a subject first
Advertisements
Advertisements
Which of the following statements is true?
Concept: undefined >> undefined
Which of the following analogies is correct?
Concept: undefined >> undefined
Advertisements
Match the items given in Column I with the items given in Column II.
| Column I | Column II |
| (i) Mg in solid state | (a) p-Type semiconductor |
| (ii) MgCl2 in molten state | (b) n-Type semiconductor |
| (iii) Silicon with phosphorus | (c) Electrolytic conductors |
| (iv) Germanium with boron | (d) Electronic conductors |
Concept: undefined >> undefined
Consider a first order gas phase decomposition reaction given below :
\[\ce{A(g) -> B(g) + C(g)}\]
The initial pressure of the system before decomposition of A was pi. After lapse of time ‘t’, total pressure of the system increased by x units and became ‘pt’ The rate constant k for the reaction is given as ______.
Concept: undefined >> undefined
Which of the following statements is not correct about order of a reaction.
Concept: undefined >> undefined
Rate law for the reaction \[\ce{A + 2B -> C}\] is found to be Rate = k [A][B]. Concentration of reactant ‘B’ is doubled, keeping the concentration of ‘A’ constant, the value of rate constant will be ______.
Concept: undefined >> undefined
Compounds ‘A’ and ‘B’ react according to the following chemical equation.
\[\ce{A(g) + 2B(g) -> 2C(g)}\]
Concentration of either ‘A’ or ‘B’ were changed keeping the concentrations of one of the reactants constant and rates were measured as a function of initial concentration. Following results were obtained. Choose the correct option for the rate equations for this reaction.
| Experiment | Initial concentration of [A]/mol L–¹ |
Initial concentration of [B]/mol L–¹ |
Initial rate of formation of [C]/mol L–¹ s–¹ |
| 1. | 0.30 | 0.30 | 0.10 |
| 2. | 0.30 | 0.60 | 0.40 |
| 3. | 0.60 | 0.30 | 0.20 |
Concept: undefined >> undefined
The value of rate constant of a pseudo first order reaction ______.
Concept: undefined >> undefined
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
Concept: undefined >> undefined
In any unimolecular reaction:
(i) only one reacting species is involved in the rate determining step.
(ii) the order and the molecularity of slowest step are equal to one.
(iii) the molecularity of the reaction is one and order is zero.
(iv) both molecularity and order of the reaction are one.
Concept: undefined >> undefined
For a complex reaction:
(i) order of overall reaction is same as molecularity of the slowest step.
(ii) order of overall reaction is less than the molecularity of the slowest step.
(iii) order of overall reaction is greater than molecularity of the slowest step.
(iv) molecularity of the slowest step is never zero or non interger.
Concept: undefined >> undefined
For which type of reactions, order and molecularity have the same value?
Concept: undefined >> undefined
In a reaction if the concentration of reactant A is tripled, the rate of reaction becomes twenty seven times. What is the order of the reaction?
Concept: undefined >> undefined
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?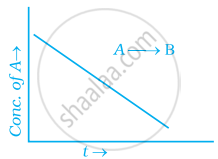
Concept: undefined >> undefined
Why is the probability of reaction with molecularity higher than three very rare?
Concept: undefined >> undefined
Why does the rate of any reaction generally decreases during the course of the reaction?
Concept: undefined >> undefined
Why can’t molecularity of any reaction be equal to zero?
Concept: undefined >> undefined
Why molecularity is applicable only for elementary reactions and order is applicable for elementary as well as complex reactions?
Concept: undefined >> undefined
Why can we not determine the order of a reaction by taking into consideration the balanced chemical equation?
Concept: undefined >> undefined
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
Concept: undefined >> undefined
