Please select a subject first
Advertisements
Advertisements
Write the structures of the following molecule:
H2SO3
Concept: Group 16 Elements - The Oxygen Family >> Oxoacids of Sulphur
Give reasons for the following : H2Te is the strongest reducing agent amongst all the hydrides of Group 16 elements.
Concept: Group 16 Elements - The Oxygen Family
Give reasons for the following : Phenol is more acidic than ethanol.
Concept: Chemical Properties of Alcohols and Phenols
Give reasons for the following : Boiling point of ethanol is higher in comparison to methoxymethane.
Concept: Chemical Properties of Alcohols and Phenols
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Concept: Methods of Preparation of Alcohols
Draw the structures of the following:
(1) HClO4
(2) H3PO3
Concept: Group 17 Elements - The Halogen Family >> Oxoacids of Halogens
Complete the following equation:
\[\ce{XeF2 + H2O ->}\]
Concept: Group 18 Elements - The Noble gas Family
Give a simple chemical test to distinguish between the following pair of compounds:
Ethanal and Propanal
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Oxidation
A compound 'A' of molecular formula C2H3OCl undergoes a series of reactions as shown below. Write the structures of A, B, C and D in the following reactions :

Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Distinguish between the following : Benzoic acid and methyl benzoate
Concept: Acids >> Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of O-H Bond
Write the structure of 2-methylbutanal.
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Nomenclature of Aldehydes and Ketones
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
Zn − Hg/conc. HCl
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Reduction
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
H2N − NHCONH2/H+
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Arrange the following in the increasing order of their boiling points : C2H5OH, CH3 − CHO, CH3 − COOH
Concept: Nature of Carbonyl Group
Give a simple chemical test to distinguish between the following pair of compounds :
CH3CH2CHO and CH3CH2COCH3
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Oxidation
How will you bring about the following conversion?
Benzoyl chloride to benzaldehyde.
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Preparation of Aldehydes
Describe the following:
Cannizzaro reaction
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Chemical Reactions of Aldehydes and Ketones - Other Reactions
Complete the synthesis by giving missing starting material, reagent or product.
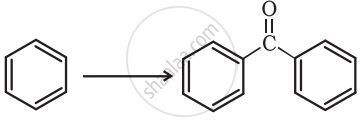
Concept: Concepts of Aldehydes, Ketones, and Carboxylic Acids >> Preparation of Ketones
Arrange the following in increasing order of their basic strength in aqueous solution:
\[\ce{CH3NH2, (CH3)3N, (CH3)2NH}\]
Concept: Concept of Amines >> Chemical Reactions of Amines - Basic Character of Amines
Arrange the following in increasing order of their basic strength :
C6H5 – NH2, C6H5 – CH2 – NH2, C6H5 – NH – CH3
Concept: Physical Properties of Amines
