Advertisements
Advertisements
ASSERTION (A): The electrical conductivity of a semiconductor increases on doping.
REASON (R): Doping always increases the number of electrons in the semiconductor.
Concept: Classification of Metals, Conductors and Semiconductors
The figure shows a piece of pure semiconductor S in series with a variable resistor R and a source of constant voltage V. Should the value of R be increased or decreased to keep the reading of the ammeter constant, when semiconductor S is heated? Justify your answer

Concept: Extrinsic Semiconductor
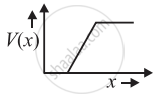
The graph of potential barrier versus width of depletion region for an unbiased diode is shown in graph A. In comparison to A, graphs B and C are obtained after biasing the diode in different ways. Identify the type of biasing in B and C and justify your answer
| ‘A’ | ‘B’ | ‘C’ |
 |
 |
 |
Concept: Semiconductor Diode
Read the following paragraph and answer the questions.
|
LED is a heavily doped P-N junction which under forward bias emits spontaneous radiation. When it is forward-biased, due to recombination of holes and electrons at the junction, energy is released in the form of photons. In the case of Si and Ge diode, the energy released in recombination lies in the infrared region. LEDs that can emit red, yellow, orange, green and blue light are commercially available. The semiconductor used for fabrication of visible LEDs must at least have a band gap of 1.8 eV. The compound semiconductor Gallium Arsenide – Phosphide is used for making LEDs of different colours.
|
- Why are LEDs made of compound semiconductor and not of elemental semiconductors?
- What should be the order of bandgap of an LED, if it is required to emit light in the visible range?
- A student connects the blue coloured LED as shown in the figure. The LED did not glow when switch S is closed. Explain why?

OR
iii. Draw V-I characteristic of a p-n junction diode in
(i) forward bias and (ii) reverse bias
Concept: Special Purpose P-n Junction Diodes
With the help of a circuit diagram, explain briefly how a p-n junction diode works as a half-wave rectifier.
Concept: Application of Junction Diode as a Rectifier
Draw V-I characteristics of a p-n Junction diode.
Concept: Semiconductor Diode
Differentiate between the threshold voltage and the breakdown voltage for a diode.
Concept: Semiconductor Diode
Two crystals C1 and C2, made of pure silicon, are doped with arsenic and aluminium respectively.
Identify the extrinsic semiconductors so formed.
Concept: Extrinsic Semiconductor
Two crystals C1 and C2, made of pure silicon, are doped with arsenic and aluminium respectively.
Why is doping of intrinsic semiconductors necessary?
Concept: Intrinsic Semiconductor
Give two differences between a half-wave rectifier and a full-wave rectifier.
Concept: Application of Junction Diode as a Rectifier
Briefly explain how emf is generated in a solar cell.
Concept: Special Purpose P-n Junction Diodes
Draw solar cells of I-V characteristics.
Concept: Special Purpose P-n Junction Diodes
Draw the circuit diagram of an illuminated photodiode and its I-V characteristics.
Concept: Special Purpose P-n Junction Diodes
How can a photodiode be used to measure light intensity?
Concept: Special Purpose P-n Junction Diodes
Name the extrinsic semiconductors formed when pure germanium is doped with a trivalent impurity. Draw the energy band diagram of extrinsic semiconductors so formed.
Concept: Extrinsic Semiconductor
Name the extrinsic semiconductors formed when pure germanium is doped with a Pentavalent impurity. Draw the energy band diagram of extrinsic semiconductors so formed.
Concept: Extrinsic Semiconductor
A semiconductor device is connected in series with a battery, an ammeter and a resistor. A current flows in the circuit. If. the polarity of the battery is reversed, the current in the circuit almost becomes zero. The device is a/an ______.
Concept: Semiconductor Diode
With the help of a circuit diagram, explain how a full wave rectifier gives output rectified voltage corresponding to both halves of the input ac voltage.
Concept: Application of Junction Diode as a Rectifier
- Assertion (A): The resistance of an intrinsic semiconductor decreases with an increase in its temperature.
- Reason (R): The number of conduction electrons as well as hole increase in an intrinsic semiconductor with the rise in its temperature.
Concept: Intrinsic Semiconductor
- Assertion (A): Putting the p-type semiconductor slab directly in physical contact with the n-type semiconductor slab cannot form the pn junction.
- Reason (R): The roughness at contact will be much more than inter atomic crystal spacing and continuous flow of charge carriers is not possible.
Concept: Extrinsic Semiconductor

