Advertisements
Advertisements
Two nuclei have mass numbers in the ratio 1: 2. What is the ratio of their nuclear densities?
Concept: Atomic Masses and Composition of Nucleus
A radioactive nucleus ‘A’ undergoes a series of decays according to the following scheme:
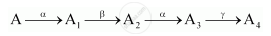
The mass number and atomic number of A are 180 and 72 respectively. What are these numbers for A4?
Concept: Law of Radioactive Decay
The radioactive isotope D decays according to the sequence
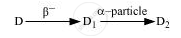
If the mass number and atomic number of D2 are 176 and 71 respectively, what is (i) the mass number (ii) atomic number of D?
Concept: Law of Radioactive Decay
Which property of nuclear force explains the constancy of binding energy per nucleon `((BE)/A)` for nuclei in the range 20< A < 170 ?
Concept: Mass-energy and Nuclear Binding Energy >> Nuclear Binding Energy
Define the term 'decay constant' of a radioactive sample. The rate of disintegration of a given radioactive nucleus is 10000 disintegrations/s and 5,000 disintegrations/s after 20 hr. and 30 hr. respectively from start. Calculate the half-life and the initial number of nuclei at t= 0.
Concept: Law of Radioactive Decay
Write two distinguishing features of nuclear forces.
Concept: Nuclear Force
Complete the following nuclear reactions for α and β deca :
(i) `"_92^238U ->? + _2^4He + Q`
(ii) `"_11^22Na ->? + _10^22Ne + v`
Concept: Forms of Energy > Nuclear Energy >> Nuclear Reactor
Determine the distance of the closest approach when an alpha particle of kinetic energy 4.5 MeV strikes a nucleus of Z = 80, stops and reverses its direction.
Concept: Radioactivity >> Alpha Decay
The figure shows the plot of binding energy (BE) per nucleon as a function of mass number A. The letters A, B, C, D, and E represent the positions of typical nuclei on the curve. Point out, giving reasons, the two processes (in terms of A, B, C, D, and E ), one of which can occur due to nuclear fission and the other due to nuclear fusion.

Concept: Mass-energy and Nuclear Binding Energy >> Nuclear Binding Energy
Identify the nature of the radioactive radiations emitted in each step of the decay process given below.
`""_Z^A X -> _Z^A _-1^-4 Y ->_Z^A _-1^-4 W`
Concept: Law of Radioactive Decay
A radioactive substance disintegrates into two types of daughter nuclei, one type with disintegration constant λ1 and the other type with disintegration constant λ2 . Determine the half-life of the radioactive substance.
Concept: Law of Radioactive Decay
Answer the following question.
Draw the curve showing the variation of binding energy per nucleon with the mass number of nuclei. Using it explains the fusion of nuclei lying on the ascending part and fission of nuclei lying on the descending part of this curve.
Concept: Mass-energy and Nuclear Binding Energy >> Nuclear Binding Energy
James Chadwick, in 1932 studied the emission of neutral radiations when Beryllium nuclei were bombarded with alpha particles. He concluded that emitted radiations were neutrons and not photons. Explain.
Concept: Atomic Masses and Composition of Nucleus
Two nuclei may have the same radius, even though they contain different numbers of protons and neutrons. Explain.
Concept: Atomic Masses and Composition of Nucleus
The radius of \[\ce{^27_13X}\] nucleus is R. The radius of \[\ce{^125_53Y}\] nucleus will be ______.
Concept: Size of the Nucleus
Calculate the binding energy of an alpha particle in MeV. Given
mass of a proton = 1.007825 u
mass of a neutron = 1.008665 u
mass of He nucleus = 4.002800 u
1u = 931 MeV/c2
Concept: Mass-energy and Nuclear Binding Energy >> Nuclear Binding Energy
The following figure shows the input waveforms (A, B) and the output waveform (Y) of a gate. Identify the gate, write its truth table and draw its logic symbol.

Concept: Digital Electronics and Logic Gates
Briefly explain how the output voltage/current is unidirectional.
Concept: Application of Junction Diode as a Rectifier
What happens when a forward bias is applied to a p-n junction?
Concept: Semiconductor Diode
When is a transistor said to be in active state?
Concept: Junction Transistor >> Transistor: Structure and Action
