Advertisements
Advertisements
Question
A radioactive nucleus ‘A’ undergoes a series of decays according to the following scheme:
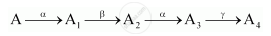
The mass number and atomic number of A are 180 and 72 respectively. What are these numbers for A4?
Advertisements
Solution
A has mass number as 180 and atomic number 172.
Formation of A1 by α-decay:
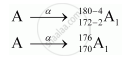
Formation of A2 by β decay:

Formation of A4:
In r-decay, mass number and atomic number remain the same.
Thus,
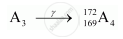
Mass number of A4 = 172
Atomic number of A4 = 169
shaalaa.com
Is there an error in this question or solution?
