Advertisements
Advertisements
Question
The radioactive isotope D decays according to the sequence
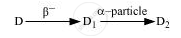
If the mass number and atomic number of D2 are 176 and 71 respectively, what is (i) the mass number (ii) atomic number of D?
Advertisements
Solution
An alpha (α) particle is a helium nucleus (`""_2^4He)`and a beta-minus decay (β−) is an emission of an electron.
The substance D2 can be represented as `""_71^176D_2`.
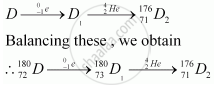
Hence,
(i) The mass number of D is 180.
(ii) The atomic number of D is 72.
shaalaa.com
Is there an error in this question or solution?
