Advertisements
Advertisements
Complete the following reaction:
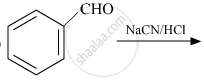
Concept: Chemical Reactions of Aldehydes and Ketones - Other Reactions
Complete the following reaction:
\[\ce{(C6H5CH2)2Cd + 2CH3COCI}\]
Concept: Preparation of Aldehydes and Ketones
Arrange the following in the increasing order of their property indicated:
Acetaldehyde, Acetone, Methyl tert butyl ketone (reactivity towards NH2OH).
Concept: Nomenclature of Aldehydes and Ketones
Give reasons to support the answer:
Presence of Alpha hydrogen in aldehydes and ketones is essential for aldol condensation.
Concept: Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Write the IUPAC name for the following organic compound:
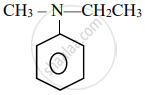
Concept: Nomenclature of Aldehydes and Ketones
Complete the following:
\[\ce{C6H5NO2 ->[Sn/HCl] A ->[Br2/H2O] B ->[NaNO2/HCl][273 - 278 K] C ->[HBF4][Δ] D}\]
Concept: Nomenclature of Carboxylic Acids
Which of the following tests/reactions is given by aldehydes as well as ketones?
Concept: Chemical Reactions of Aldehydes and Ketones - Oxidation
Write the reaction and IUPAC name of the product formed when 2-Methylpropanal (isobutyraldehyde) is treated with ethyl magnesium bromide followed by hydrolysis.
Concept: Nomenclature of Aldehydes and Ketones
Write the equations for the following reaction:
Salicylic acid is treated with acetic anhydride in the presence of conc H2SO4
Concept: Chemical Reactions of Carboxylic Acids - Reactions Involving Cleavege of C-OH Bond
A hydrocarbon (A) with molecular formula C5H10 on ozonolysis gives two products (B) and (C). Both (B) and (C) give a yellow precipitate when heated with iodine in presence of NaOH while only (B) give a silver mirror on reaction with Tollen’s reagent.
- Identify (A), (B) and (C).
- Write the reaction of B with Tollen’s reagent.
- Write the equation for iodoform test for C.
- Write down the equation for aldol condensation reaction of B and C.
Concept: Chemical Reactions of Aldehydes and Ketones - Oxidation
Arrange the following in the increasing order of their property indicated:
Ethanal, Propanone, Propanal, Butanone (reactivity towards nucleophilic addition)
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
Write the IUPAC name of the following complex:
K2[PdCl4]
Concept: Nomenclature of Aldehydes and Ketones
Explain the following reactions:
Clemmensen reaction
Concept: Chemical Reactions of Aldehydes and Ketones - Reduction
Write the IUPAC name of the following complex:
[Pt(NH3)6]Cl4
Concept: Nomenclature of Aldehydes and Ketones
Using IUPAC norms, write the formula for the following:
Pentaamminenitrito-N-Cobalt (III)
Concept: Nomenclature of Aldehydes and Ketones
Using IUPAC norms write the formula for the following:
Tetrahydroxidozincate (II)
Concept: Nomenclature of Aldehydes and Ketones
Predict the reagent for carrying out the following transformations:
Ethanal to 3-hydroxy butanal
Concept: Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Which of the following does not give aldol condensation reaction?
Concept: Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
Which of the following does not give Cannizzaro reaction?
Concept: Chemical Reactions of Aldehydes and Ketones - Other Reactions
Aldehydes and ketones react with hydroxylamine to form ______.
Concept: Chemical Reactions of Aldehydes and Ketones - Nucleophilic Addition Reactions
