Definitions [20]
Primary amines (1°) are the ammonia derivatives in which one H-atom has been replaced by one alkyl or aryl group.
Tertiary amines (3°) are the ammonia derivatives in which three hydrogen atoms have been replaced by three alkyl or aryl group.
Secondary amines (2°) are the ammonia derivatives in which two hydrogen atoms have been replaced by two alkyl or aryl groups.
The general formula for diazonium salts is ArN₂⁺X⁻, where Ar refers to an aryl group while X⁻ ion may be Cl⁻, Br⁻, HSO₄⁻, BF₄⁻, etc.; the group N₂⁺ or [—N≡N] is called the diazo group.
Benzene diazonium chloride reacts with phenol in which phenol molecule at its para-position is coupled with diazonium salt is give the product p-hydroxy azobenzene. Hence, the reaction is known as coupling reaction.
An amine in which two hydrogen atoms of ammonia are replaced by alkyl or aryl groups is called secondary amine.
R–NH–R′
The substitution reaction in which diazonium group is replaced by Cl, Br or CN in the presence of Cu(I) salts is called Sandmeyer reaction.
\[ArN_2^+Cl^-\xrightarrow{CuCl/HCl}ArCl+N_2\]
\[ArN_2^+Cl^-\xrightarrow{CuBr/HBr}ArBr+N_2\]
\[ArN_2^+Cl^-\xrightarrow{CuCN}ArCN+N_2\]
A positively charged nitrogen species formed by addition of four alkyl/aryl groups is called quaternary ammonium salt.
R₄N⁺X⁻
The reaction in which alkyl halides react with ammonia to form amines is called ammonolysis.
The reaction of amines with nitrous acid to form diazonium salts (in case of primary aromatic amines) is called diazotization reaction.
An amine in which three hydrogen atoms of ammonia are replaced by alkyl or aryl groups is called tertiary amine.
R–N(R′)–R″
The reaction in which primary amines on heating with chloroform and alcoholic KOH form isocyanides is called Carbylamine reaction.
R – NH2 + CHCl3 + 3KOH → R – NC + 3KCl + 3H2O
The reaction of primary aromatic amines with nitrous acid at 273–278 K to form diazonium salts is called Diazotization reaction.
The reaction of primary and secondary amines with benzenesulphonyl chloride to form sulphonamides is called Hinsberg reaction.
Primary amine:
R – NH2 + C6H5SO2Cl → Sulphonamide
Secondary amine:
R2NH + C6H5SO2Cl → N,N-dialkyl sulphonamide
Tertiary amine → No reaction
The reaction in which diazonium salts couple with phenols or aromatic amines to form azo compounds is called Coupling reaction.
ArN2+ Cl− + Phenol → Azo compound
An amine in which one hydrogen atom of ammonia is replaced by an alkyl or aryl group is called primary amine.
R–NH₂
The reaction in which amines react with acid chlorides or anhydrides to form amides is called acylation.
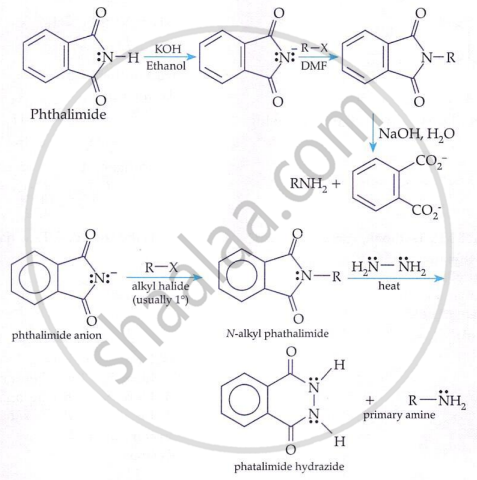
The method of preparation of primary amines using potassium phthalimide is called Gabriel phthalimide synthesis.
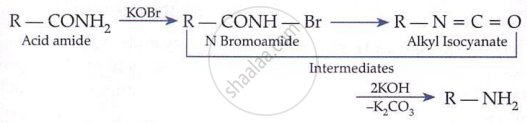
The degradation of amides to primary amines containing one carbon less is called Hofmann bromamide degradation reaction.
Organic compounds derived from ammonia by replacement of one or more hydrogen atoms by alkyl or aryl groups are called amines.
Chemica Equations [7]
\[R–NO_2\xrightarrow{H_2/Pd}R–NH_2\]
\[R\neg CN\xrightarrow{LiAlH_4}R\neg CH_2NH_2\]
R – CONH2 + Br2 + 4NaOH → R – NH2 + Na2CO3 + 2NaBr + 2H2O
R – X + NH3 → R – NH2 + HX
Salt + R – X → R – NH2
R – NH2 + R′COCl → R – NHCO – R′ + HCl
Ar – NH2 + NaNO2 + HCl → Ar – N2+ Cl−
Key Points
- Amines overview: Amines are derivatives of ammonia (NH₃) where one or more H-atoms are replaced by alkyl/aryl groups; they can be saturated or unsaturated.
- Classification by source: Aliphatic amines (e.g., CH₃–NH₂, methylamine) and Aromatic amines (e.g., C₆H₅–NH₂, aniline).
- Classification by H-substitution: Primary (1°, –NH₂, e.g., methylamine), Secondary (2°, –NH–, e.g., dimethylamine), and Tertiary (3°, –N–, e.g., trimethylamine).
- Sub-types of 2° and 3° amines: Simple/symmetrical (same groups, e.g., (C₂H₅)₂NH, (C₂H₅)₃N) and Mixed/unsymmetrical (different groups, e.g., C₂H₅–NH–CH₃, C₂H₅–N(CH₃)₂).
- Examples of each class: 1° – n-butylamine, sec-butylamine, tert-butylamine, aniline; 2° – dimethylamine, diethylamine, diphenylamine; 3° – trimethylamine, triethylamine, triphenylamine.
- Common system: Aliphatic amine is named by adding 'amine' to the alkyl group (e.g., CH₃NH₂ – methylamine).
- Same groups in 2°/3° amines: Use prefix di or tri before the alkyl group name (e.g., trimethylamine).
- IUPAC – Primary amines: Named as alkanamines, replacing 'e' of alkane with 'amine' (e.g., methanamine).
- IUPAC – 2° & 3° amines: Named as N-alkylalkanamine or N,N-dialkylalkanamine (e.g., N,N-dimethylmethanamine).
- Multiple —NH₂ groups: Positions are numbered, terminal 'e' retained (e.g., ethane-1,2-diamine); aromatic amines named as aniline derivatives (e.g., N, N-dimethylaniline).
Replace 'e' of alkane by amine'.
For example, \[ \underset{\text{Ethanamine}}{\mathrm{CH}_3\mathrm{CH}_2\mathrm{NH}_2} \quad , \quad \underset{\text{Propan-1-amine}}{\mathrm{CH}_3\mathrm{CH}_2\mathrm{CH}_2\mathrm{NH}_2} \]
Secondary amines are named as N-alkylaminoalkanes, and tertiary amines are named as N, N-dialkylaminoalkanes
For example,
\[ \underset{\text{N-methylethanamine}}{\mathrm{CH}_3 - \underset{\underset{\displaystyle \mathrm{H}}{|}}{\mathrm{N}} - \mathrm{CH}_2 - \mathrm{CH}_3} \quad , \quad \underset{\text{N-methylmethanamine}}{\mathrm{CH}_3 - \underset{\underset{\displaystyle \mathrm{H}}{|}}{\mathrm{N}} - \mathrm{CH}_3} \]
Aromatic amine,
It is named as aniline or benzenamine.
Reduction of Alkyl Nitriles (Nitrile Reduction):
\[ \mathrm{R} - \mathrm{C} \equiv \mathrm{N} \xrightarrow[\mathrm{LiAlH}_4 \text{ or } \mathrm{Na} / \mathrm{C}_2\mathrm{H}_5\mathrm{OH}]{\text{Raney Ni} / \mathrm{H}_2} \mathrm{R} - \mathrm{CH}_2\mathrm{NH}_2 \]
Reduction of Amides:
\[ \mathrm{R} - \mathrm{CONH}_2 \xrightarrow{\mathrm{LiAlH}_4 / \text{ether}} \mathrm{R} - \mathrm{CH}_2\mathrm{NH}_2 \]
Reduction of Oximes:
\[ \mathrm{R} - \mathrm{CH} = \mathrm{NOH} + 4[\mathrm{H}] \xrightarrow[\mathrm{LiAlH}_4]{\mathrm{Na} / \mathrm{C}_2\mathrm{H}_5\mathrm{OH}} \mathrm{R} - \mathrm{CH}_2\mathrm{NH}_2 + \mathrm{H}_2\mathrm{O} \]
Reductive Amination (from Aldehydes/Ketones):
\[ \mathrm{R} - \mathrm{CH} = \mathrm{O} \xrightarrow{\mathrm{NH}_2\mathrm{OH}} \mathrm{R} - \mathrm{CH} = \mathrm{N} - \mathrm{OH} \xrightarrow{\mathrm{LiAlH}_4} \mathrm{R} - \mathrm{CH}_2 - \mathrm{NH}_2 \]
Gabriel Phthalimide Reaction (for 1° amines only):
Hoffmann Bromamide Degradation (gives 1° amine with one less carbon):
\[ \underset{\text{Amide}}{\mathrm{RCONH}_2} + \mathrm{Br}_2 + 4\mathrm{KOH} \longrightarrow \underset{1^\circ \text{ amine}}{\mathrm{RNH}_2} + 2\mathrm{KBr} + 2\mathrm{H}_2\mathrm{O} + \mathrm{K}_2\mathrm{CO}_3 \]
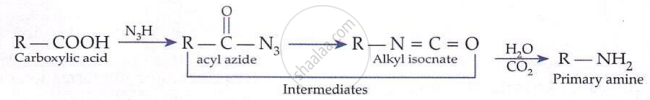
Mechanism:
- Boiling point vs non-polar compounds: Due to intermolecular H-bonding, amines boil at a higher temperature than non-polar compounds such as hydrocarbons.
- Boiling point vs alcohols/acids: The boiling points of amines are less than those of alcohols and carboxylic acids.
- Solubility of lower amines: Lower members of aliphatic amines are soluble in water due to intermolecular H-bonding with water.
- Effect of alkyl group size: Solubility in water decreases as the size of the alkyl group increases.
- Order of boiling points & aromatic amines: Propane < ethylamine < diethylamine < n-butylamine < ethyl alcohol < propanoic acid; aromatic amines are insoluble in water due to the bulky size of the phenyl group.
- Basic nature: Amines are basic in nature, so they react with acids and form salts.
- Basicity criterion: The larger the value of K_b or the smaller the value of pK_b, the stronger the base.
- Order of pK_b values: NH₃ > R—NH₂ > R₂NH > R₃N.
- Order of basic strength (aqueous): NH₃ < R—NH₂ < R₂NH < R₃N; in gaseous phase the order is 3° amine > 2° amine > 1° amine > NH₃.
- Substituent effect on aniline: Electron-donating groups (—CH₃, —OCH₃) increase basicity, while electron-withdrawing groups (—CHO, —NO₂) decrease the basicity of aniline.
- Hoffmann's exhaustive alkylation: R—NH₂ + R—X → successively gives 2°, 3° amines, and finally tetraalkyl ammonium halide (R₄N⁺X⁻).
- Hoffmann elimination: Quaternary ammonium hydroxide on heating with moist Ag₂O gives an alkene + 3° amine + H₂O.
- Acylation: Amines + CH₃COCl (in pyridine) → N-substituted amide + HCl.
- Carbylamine reaction: R—NH₂ + CHCl₃ + 3KOH → R—NC (alkyl isocyanide) + 3KCl + 3H₂O.
- Reaction with HNO₂ (273–278 K): Aliphatic 1° amine → R—OH + N₂ + HCl; aromatic 1° amine → diazonium salt (C₆H₅—N⁺≡NCl⁻).
- Preparation: Primary aromatic amines (aniline) react with HNO₂ (NaNO₂ + 2HCl) at 273–278 K (0–5 °C) to form diazonium chloride + NaCl + 2H₂O.
- Sandmeyer reactions: C₆H₅N₂⁺Cl⁻ with CuCl/HCl, CuBr/HBr, CuCN/KCN give C₆H₅Cl, C₆H₅Br, C₆H₅CN, respectively (with N₂↑).
- Other displacement reactions: Gattermann (Cu/HCl) → C₆H₅Cl; KI → C₆H₅I; H₃PO₂/H₂O → C₆H₆; H₂O/283 K → C₆H₅OH; HBF₄/Δ → C₆H₅F (Balz–Schiemann reaction).
- Coupling with phenol: Benzene diazonium chloride + phenol (OH⁻) → p-hydroxy azobenzene (orange dye) + HCl.
- Coupling with aniline: Benzene diazonium chloride + aniline (mild alkaline medium) → p-aminoazobenzene (yellow dye) + HCl; azo products are coloured and used as dyes.
- Hinsberg's reagent is arene sulphonyl chloride (C₆H₅SO₂Cl).
- This test is used to distinguish three classes of amines, i.e., 1°, 2° and 3° amines.
- With a 1° amine (RNH₂), C₆H₅SO₂Cl gives C₆H₅SO₂NH—R (with loss of HCl), which is soluble in alkali.
- With a 2° amine (R₂NH), it gives C₆H₅SO₂NR₂ (with loss of HCl), which is insoluble in alkali.
- With a 3° amine (R₃N), there is no reaction.
- Bromination: Aniline reacts with Br₂/H₂O to give 2,4,6-tribromoaniline + 3HBr (all three ortho/para positions substituted).
- Nitration: Aniline with conc. HNO₃ + conc. H₂SO₄ at 288 K gives p-nitroaniline (51%), m-nitroaniline (47%) and o-nitroaniline (2%).
- Getting p-nitroaniline as a major product: —NH₂ is first protected by acetylation, then nitration is carried out, and finally the amide is hydrolysed.
- Sulphonation: Aniline + H₂SO₄ first forms anilinium hydrogen sulphate, which on heating at 453–473 K gives sulphanilic acid (47%).
- Zwitter ion: Sulphanilic acid exists in equilibrium with its dipolar (zwitter ion) form (2%) carrying —NH₃⁺ and —SO₃⁻ groups.
Primary amides on treatment with bromine and sodium hydroxide give primary amines containing one carbon atom less than the parent amide.
Key Points:
-
Carbonyl carbon is lost as CO₂.
-
Rearrangement reaction.
-
Useful for chain shortening.
Primary and secondary amines react with acid chlorides or anhydrides to form amides via nucleophilic substitution.
Key Points:
-
Used for identification of amines.
-
Tertiary amines do not undergo acylation.
-
Reaction carried out in presence of base.
Amines are basic in nature due to the presence of a lone pair of electrons on nitrogen which can accept a proton.
Factors affecting basicity:
- +I effect of alkyl groups increases basicity.
- Solvation effect in aqueous solution.
- Steric hindrance decreases basicity.
- Aromatic amines are less basic due to resonance.
Order in gaseous phase:
3∘ > 2∘ >1∘ >NH3
Order in aqueous phase:
2∘ > 1∘ > 3∘ > NH3
Primary amines are prepared by treating potassium phthalimide with alkyl halides followed by hydrolysis.
Key points:
-
Gives only primary amines.
-
Not suitable for aryl halides.
-
Avoids formation of secondary and tertiary amines.
Important Questions [18]
- The Compound ‘B’ is
- Which Among the Following Molecular Formulae Represents Urotropine?
- How are ethylamine and ethyl methyl amine distinguished by using nitrous acid?
- What is the Action of P-toluenesulphonychloride on Ethylamine and Diethylamine ?
- Write the chemical equation involved in the following reaction: Hoffmann-bromamide degradation reaction
- Write a short note on the following: Hoffmann’s bromamide reaction
- Explain the Mechanism of Action of Hydroiodic Acid on 3-methylbutan-2-ol.
- Mention 'Two' Uses of Propan-2-one.
- Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
- How Are Propan-1-amine and Propan-2-amine Prepared from Oxime?
- Write a Short Note on Hoffmann Bromamide Degradation.
- Identify the compounds 'A' and 'B' in the following equation
- Write the name of the product formed by the action of LiAlH4/ether on acetamide.
- How is Ethyl Amine Prepared from Methyl Iodide?
- Identify 'A' and 'B' in the Following Reaction and Rewrite the Complete Reaction :
- Write reactions to bring about the following conversions. Acetamide to Ethylamine
- Identify the Weakest Base Amongst the Following
- Write a short note on Hofmann elimination
