Definitions [49]
Organic compounds containing carbon-oxygen double bond, i.e. \[\mathrm{>C=O}\] group, are known as carbonyl compounds.
The organic compounds formed by removal of one molecule of water from two molecules of carboxylic acid are called acid anhydrides.
The organic compounds in which the –OH group of a carboxylic acid is replaced by –NH₂ or substituted amino group are called amides.
The carbon–oxygen double bond (>C=O) functional group present in aldehydes, ketones and acids is called carbonyl group.
The organic compounds in which the –OH group of a carboxylic acid is replaced by an –OR group are called esters.
The organic compounds in which the –OH group of a carboxylic acid is replaced by a halogen atom are called acyl halides.
Organic compounds in which the carbonyl group is attached to two alkyl or aryl groups are called ketones (>C=O).
Ketones having different groups attached to the carbonyl carbon.
Organic compounds in which the carbonyl group is attached to at least one hydrogen atom are called aldehydes (–CHO group).
Aldehydes in which the –CHO group is attached to an alkyl group or hydrogen are called aliphatic aldehydes.
Aldehydes in which the –CHO group is attached to an aryl group are called aromatic aldehydes.
Ketones in which both groups attached to the carbonyl carbon are alkyl groups.
The unstable addition product formed when aldehydes react with ammonia is called aldehyde ammonia.
A compound containing the functional group –C=NH formed by dehydration of aldehyde ammonia is called an aldimine (Schiff base).
The compound formed by the reaction of aldehydes or ketones with hydroxylamine having the group >C=NOH is called an oxime.
The derivative formed when aldehydes or ketones react with phenylhydrazine is called a phenylhydrazone.
Ketones in which one or both groups attached to the carbonyl carbon are aryl groups.
The disproportionation reaction of aldehydes without α-hydrogen atoms in concentrated alkali is called the Cannizzaro reaction.
The dehydration of aldol to form an α,β-unsaturated carbonyl compound is called crotonisation.
The condensation reaction of aldehydes or ketones containing α-hydrogen atoms in dilute alkali to form β-hydroxy compounds is called aldol condensation.
The test in which the pink colour of Schiff’s reagent is restored by aldehydes but not by ketones is called Schiff’s test.
The electrophilic substitution reaction in which aromatic aldehydes or ketones react with nitric acid in the presence of sulphuric acid to form nitro compounds is called nitration.
The electrophilic substitution reaction in which aromatic aldehydes or ketones react with concentrated sulphuric acid to introduce a sulphonic acid group is called sulphonation.
The compound formed by the reaction of aldehydes or ketones with semicarbazide is called a semicarbazone.
The oxidation of alkyl side chains of aromatic hydrocarbons to –COOH group is called side-chain oxidation.
The compound formed by the reaction of aldehydes or ketones with hydrazine having the group >C=NNH₂ is called a hydrazone.
The process of converting primary alcohols or aldehydes into carboxylic acids using oxidising agents is called oxidation.
Compounds containing the –C≡N (cyanide) group are called nitriles.
The preparation of carboxylic acids by hydrolysis of acid chlorides or acid anhydrides is called acyl hydrolysis.
Ketones having identical groups attached to the carbonyl carbon.
The reduction of aldehydes and ketones to hydrocarbons using zinc amalgam and concentrated hydrochloric acid is called Clemmensen reduction.
The reduction of aldehydes and ketones to hydrocarbons using hydrazine and strong alkali at high temperature is called Wolff–Kishner reduction.
The test in which aldehydes reduce Tollens’ reagent to metallic silver producing a silver mirror is called Tollens’ test.
The test in which aliphatic aldehydes reduce Fehling’s solution to a red precipitate of cuprous oxide is called Fehling’s test.
The haloform reaction carried out using iodine that gives a yellow precipitate of iodoform is called the iodoform test.
The reaction in which an aromatic aldehyde reacts with an acid anhydride in the presence of sodium salt of the corresponding acid to form an α,β-unsaturated carboxylic acid is called Perkin reaction.
The substitution reaction in which aromatic aldehydes or ketones react with halogens to form halo-substituted products is called halogenation.
The functional group formed by the combination of a carbonyl group (–C=O) and a hydroxyl group (–OH) is called the carboxyl group.
A carboxylic acid in which the –COOH group is attached directly to an aryl group (benzene ring) is called an aromatic carboxylic acid.
A carboxylic acid containing only one carboxyl group in its molecule is called a monocarboxylic acid.
A carboxylic acid containing two carboxyl groups in its molecule is called a dicarboxylic acid.
A carboxylic acid containing three carboxyl groups in its molecule is called a tricarboxylic acid.
Saturated aliphatic monocarboxylic acids having 12–18 carbon atoms and occurring naturally in fats and oils are called fatty acids.
A carboxylic acid in which the –COOH group is attached to a hydrogen atom or an alkyl group is called an aliphatic carboxylic acid.
The chemical combination of a large number of similar or identical small molecules (monomers) to form a high molecular mass compound is called polymerisation.
The self-condensation reaction of aromatic aldehydes in the presence of alcoholic potassium cyanide to form α-hydroxy ketones is called benzoin condensation.
An organic compound containing the carboxyl group (–COOH) as its functional group is called a carboxylic acid.
The functional group >C=O present in aldehydes and ketones is called the carbonyl group.
Organic compounds containing the carbonyl group (>C=O) are collectively called carbonyl compounds.
Formulae [4]
R–COX
R–COOR′
(R–CO)₂O
R–CONH₂
Chemica Equations [5]
\[2CH_3CHO\xrightarrow{dil.NaOH}CH_3CH(OH)CH_2CHO\]
On heating:
→CH3CH = CHCHO + H2O
\[RCOOH+R^{\prime}OH\xrightarrow{H^+}RCOOR^{\prime}+H_2O\]
\[RCHO\xrightarrow{NaBH_4}RCH_2OH\]
\[RCOOH\xrightarrow{LiAlH_4}RCH_2OH\]
\[RCOONa+NaOH\xrightarrow{CaO}RH+Na_2CO_3\]
Key Points
- Carbonyl group: The ≻C=O group (carbonyl carbon + carbonyl oxygen) — a key functional group in organic chemistry.
- Carbonyl compounds: Aldehydes and ketones, both containing ≻C=O as their functional group.
- Aldehydes: –CHO (formyl group); carbonyl C bonded to at least one H.
- Ketones: ≻C=O (ketonic carbonyl group); carbonyl C bonded to two alkyl/aryl groups (R=R′ or R≠R′).
- Carboxylic acids: –COOH (carboxyl group); –OH attached to ≻C=O makes them distinct from aldehydes/ketones.
- Ether C–O cleavage reactions: With HX (100 °C) → R–OH + R–X; PCl₅ → R–Cl + R′–Cl; dil. H₂SO₄ → R–OH + R′–OH; R′COCl/(R′CO)₂O with AlCl₃ → esters.
- Reactivity order of HX with ethers: HI > HBr > HCl.
- Preparation of ethers: Dehydration of alcohols (conc. H₂SO₄, 443 K), catalytic dehydration (Al₂O₃, 250 °C), Williamson synthesis (Sₙ2), and alkyl halides + dry Ag₂O.
- Carbonyl compounds classification: Aldehydes (R–CHO, Ar–CHO), ketones (aliphatic/aromatic — simple or mixed), and carboxylic acids (RCOOH, ArCOOH).
- Carboxylic acid types: Mono- (propionic acid), di- (oxalic acid), and tri-carboxylic acid (citric acid).
| Functional group | Types | IUPAC system (Basic rules) | Examples |
|---|---|---|---|
| Aldehydes | Aliphatic compounds | Parent name: Alkane → Alkanal; Prefix – Formyl | Hexanal, 3-Formylhexanoic acid |
| Alicyclic compounds | Parent name: Cycloalkanecarbaldehyde | Cyclohexanecarbaldehyde | |
| Aromatic compounds | Suffix – Benzaldehyde (one –CHO group); Prefix – Formyl | 3-Methylbenzaldehyde, 4-Formylbenzoic acid | |
| Ketones | Aliphatic compounds | Parent name: Alkane → Alkanone; Prefix – Oxo | Hexan-2-one, 3-Oxobutanal |
| Alicyclic compounds | Parent name: Cycloalkanone | 4-Methylcyclohexanone | |
| Aromatic compounds | Suffix – phenone | Benzophenone | |
| Carboxylic acids | Aliphatic compounds | Parent name: Alkane → Alkanoic acid | Hexanoic acid |
| Alicyclic compounds | Parent name: Cycloalkanecarboxylic acid | Cyclohexanecarboxylic acid | |
| Aromatic compounds | Suffix – Benzoic acid (one –COOH group) | 2-Hydroxybenzoic acid |
Preparation of aliphatic aldehydes and ketones
By oxidation of alcohols:
\[\ \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{CH}-\mathrm{OH} \\ & / & \\ \mathrm{R}' & & \end{array} + [\mathrm{O}] \xrightarrow[\text{Or KMnO}_4]{\text{K}_2\text{Cr}_2\text{O}_7/\text{H}_2\text{SO}_4} \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{C}=\mathrm{O} \\ & / & \\ \mathrm{R}' & & \end{array} + \mathrm{H}_2\mathrm{O}\]
When,
- R' = H then 1° alcohol to aldehyde.
- R' = alkyl group then 2º alcohol to ketone.
By dehydrogenation of alcohols:
\[ \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{CH}-\mathrm{OH} \\ & / & \\ \mathrm{R}' & & \end{array} \xrightarrow[\text{573 K}]{\text{Cu}} \begin{array}{r@{\;}c@{\;}l} \mathrm{R} & & \\ & \backslash & \\ & & \mathrm{C}=\mathrm{O} \\ & / & \\ \mathrm{R}' & & \end{array} + \mathrm{H}_2 \]
When
- R' = H then 1° alcohol to aldehyde.
- R' = alkyl group the 2° alcohol to ketone.
By acid chloride:
\[ \mathrm{R} - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{Cl} + \mathrm{H}_2 \xrightarrow[\text{Rosenmund Reduction}]{\text{Pd}-\text{BaSO}_4} \underset{\text{Aldehyde}}{\mathrm{R} - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{H}} + \mathrm{HCl} \]
2RMgX + CdCl2 → R2Cd + 2MgXCl
\[\ 2\mathrm{R}' - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{Cl} + \mathrm{R}_2\mathrm{Cd} \longrightarrow 2\mathrm{R}' - \underset{\text{Ketone}}{\overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}}} - \mathrm{R} + \mathrm{CdCl}_2 \]
From nitriles and esters:
\[ \mathrm{R} - \mathrm{CN} \xrightarrow[\text{(ii) }\mathrm{H}_2\mathrm{O}]{\text{(i) }\mathrm{AlH}(i\text{Bu})_2} \underset{\text{Aldehyde}}{\mathrm{R} - \mathrm{CHO}} \]
\[ \mathrm{CH}_3(\mathrm{CH}_2)_9 - \overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{OC}_2\mathrm{H}_5 \xrightarrow[\text{(ii) }\mathrm{H}_2\mathrm{O}]{\text{(i) DIBAL-H}} \mathrm{CH}_3(\mathrm{CH}_2)_9 - \underset{\text{Aldehyde}}{\overset{\displaystyle \mathrm{O}}{\overset{||}{\mathrm{C}}} - \mathrm{H}} \]
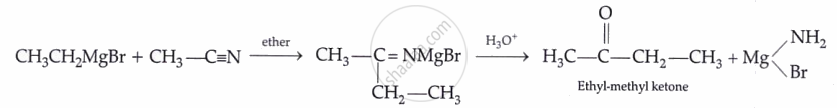 From hydrocarbons
From hydrocarbons
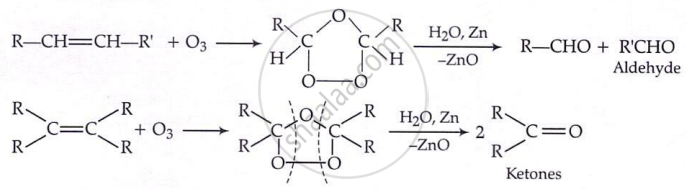
By ozonolysis:
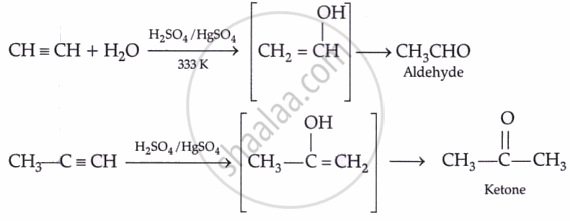
By hydration:
- Special preparations: Benzoic acid from cumene (KMnO₄/KOH, Δ → H₃O⁺); adipic acid from cyclohexene (KMnO₄/dil. H₂SO₄, Δ).
- Aldehyde preparation: From 1° alcohol (K₂Cr₂O₇/H₂SO₄ or Cu/573 K), alkene (ozonolysis), alkyne (dil. H₂SO₄/HgSO₄), acid chloride (Rosenmund), nitrile (Stephen/DIBAL-H); aromatic via Etard, CrO₃/(CH₃CO)₂O, Cl₂/hν, Gatterman–Koch.
- Aldehyde reactions: HCN → cyanohydrin; NaHSO₃ → bisulphite adduct; R′OH → acetal; RMgBr → 2° alcohol; NH₂OH → aldoxime; NH₂NH₂ → hydrazone; K₂Cr₂O₇ → COOH; Clemmensen/Wolf–Kishner → alkane.
- Carboxylic acid preparation: From nitriles (hydrolysis), acyl chloride/anhydride/ester + H₂O, CO₂ + RMgX (dry ether, H₃O⁺), alkylbenzene (KMnO₄–KOH).
- Carboxylic acid reactions: PCl₅/SOCl₂ → RCOCl; NH₃ → amide; P₂O₅ → anhydride; NaOH + CaO (Δ) → alkane; LiAlH₄ → 1° alcohol; ROH/conc. H₂SO₄ → ester.
- In the carbonyl group, the π-electron cloud is displaced towards the more electronegative oxygen, making carbon partially positive (δ+) and oxygen partially negative (δ−).
- The polarity of the carbonyl group is also explained by resonance, with electron delocalisation shown through two principal resonance forms.
- Aldehydes are more reactive than ketones towards nucleophilic attack due to two factors: electronic effect and steric effect.
- Electronic effect: Aldehydes have only one electron-donating group (EDG) attached to the carbonyl carbon, making them more electrophilic than ketones.
- Steric effect: Aldehydes have less steric hindrance than ketones, so nucleophiles can attach more easily; note that aromatic aldehydes are less reactive than aliphatic aldehydes in nucleophilic addition reactions.
- Oxidation: Aldehydes → RCOOH (same C) with dil. HNO₃/KMnO₄/K₂Cr₂O₇; Ketones undergo C–C cleavage with CrO₃, giving two carboxylic acids.
- Reduction: Clemmensen (Zn-Hg/conc. HCl) and Wolf-Kishner (NH₂-NH₂ then KOH/HOCH₂CH₂OH, Δ) both convert >C=O to >CH₂.
- Electrophilic Substitution: –CHO is EWG (inductive + resonance); deactivates ring; directs electrophile (NO₂⁺) to meta position.
- Lab Tests: Schiff (pink), Tollens (silver mirror), Fehling (red Cu₂O ppt) — for aldehydes; Sodium nitroprusside (red) — for ketones.
- Nucleophilic Reactions: HCN → cyanohydrin; NaHSO₃ → bisulphite adduct; ROH/dry HCl → hemiacetal/acetal/ketal; NaOI → haloform (CHI₃); dil. NaOH → Aldol condensation; No α-H + NaOH → Cannizzaro reaction.
- Acid strength order: Greater halogen electronegativity → stronger acid, smaller pKₐ; F–CH₂COOH > Cl–CH₂COOH > Br–CH₂COOH > I–CH₂COOH > CH₃COOH.
- Substituent effect: EWG (–Cl, –CN, –NO₂) ↑ acidity; EDG (–CH₃, –OH, –OCH₃) ↓ acidity; 4-nitrobenzoic acid > Benzoic acid > 4-methyl benzoic acid.
- Tests for –COOH: Litmus (blue → red), NaHCO₃ (CO₂↑), Ester test (with C₂H₅OH/H⁺ gives ester).
- Key conversions: SOCl₂/PCl₃/PCl₅ → acyl chloride; NH₃ (Δ) → amide; P₂O₅ (Δ) → acid anhydride.
- Decarboxylation & reduction: RCOONa + NaOH/CaO (Δ) → R–H + Na₂CO₃; RCOOH + LiAlH₄/dry ether → RCH₂OH.
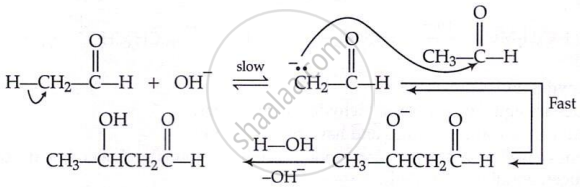
Aldol condensation:
\[\ce{CH3 - \overset{\displaystyle O}{\overset{||}{C}} - H + H - CH2 - \overset{\displaystyle O}{\overset{||}{C}} - H} \xrightarrow{\text{dil. NaOH}} \ce{H3C - \underset{\underset{\displaystyle OH}{|}}{CH} - \underset{\underset{\displaystyle H}{|}}{CH} - \overset{\displaystyle O}{\overset{||}{C}} - H} \\ \xrightarrow{\text{dil. }\ce{H2SO4}\Delta, \ce{-H2O}} \ce{CH3 - CH = CH - \overset{\displaystyle O}{\overset{||}{C}} - H}\]
Mechanism:
Cross aldol condensation:
\[\begin{array}{ccccccccccccc} & \ce{O} & & \ce{O} & & & & \ce{OH} & & \ce{O} & & & \\ & || & & || & & & & | & & || & & & \\ \ce{C6H5 -} & \ce{C} & \ce{+ HCH2 -} & \ce{C} & \ce{- H} & \overset{\text{dil. NaOH}}{\rightleftharpoons} & \ce{C6H5 -} & \ce{C} & \ce{- CH2 -} & \ce{C} & \ce{- H} & \xrightarrow[\Delta]{\ce{H2O}} & \ce{C6H5CH=CHCHO} \\ & | & & & & & & | & & & & & \text{Cinnamaldehyde} \\ & \ce{H} & & & & & & \ce{H} & & & & & \end{array}\]
Cannizzaro reaction: It is a self-oxidation reduction reaction.
\[\begin{array}{r@{\;}c@{\;}l} \ce{H} & & \\ & \backslash & \\ & & \ce{C=O} \\ & / & \\ \ce{H} & & \end{array} + \begin{array}{r@{\;}c@{\;}l} \ce{H} & & \\ & \backslash & \\ & & \ce{C=O} \\ & / & \\ \ce{H} & & \end{array} + \text{conc. KOH} \xrightarrow{\Delta} \ce{H - \underset{\underset{\displaystyle H}{|}}{\overset{\overset{\displaystyle H}{|}}{C}} - OH} + \ce{H - \overset{\displaystyle O}{\overset{||}{C}} - OK}\]
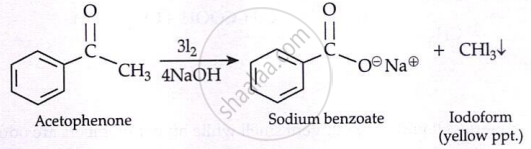
Haloform Reaction:
The reaction can be used to transform acetyl groups into carboxyl groups or to produce chloroform or iodoform. This reaction has been used in qualitative analysis to indicate the presence of a methyl ketone in which excess iodine is used to halogenate the compound. The product iodoform precipitates as a yellow-coloured substance and has a characteristic odour.
\[\underset{\substack{\text{Acetone} \\ \text{Iodoform Test}}}{\ce{H3C - \overset{\displaystyle O}{\overset{||}{C}} - CH3}} \xrightarrow[4\ce{NaOH}]{3\ce{Cl2}} \underset{\text{Sodium acetate}}{\ce{H3C - \overset{\displaystyle O}{\overset{||}{C}} - O^\ominus Na^\oplus}} + \underset{\text{Chloroform}}{\ce{CHCl3}}\]
Statement:
The carbonyl carbon atom is sp² hybridised, forms a trigonal planar structure, and possesses a polar C=O bond.
Explanation:
1. Hybridisation:
- Carbonyl carbon is sp² hybridised.
- It forms:
- Three σ (sigma) bonds.
- One π (pi) bond with oxygen.
- The π-bond is formed by sideways overlap of p-orbitals.
2. Geometry:
- The carbonyl carbon and three attached atoms lie in the same plane.
- Bond angle ≈ 120°.
- Geometry is trigonal planar.
3. Polarity:
- Oxygen is more electronegative than carbon.
- Hence, electron density shifts towards oxygen.
- Carbon acquires partial positive charge (δ⁺).
- Oxygen acquires partial negative charge (δ⁻).
4. Resonance:
Two contributing structures:
R–C=O ↔ R–C⁺–O⁻
This explains:
- High dipole moment.
- Electrophilic nature of carbonyl carbon.
Conclusion:
The planar structure, polarity and resonance make the carbonyl carbon highly reactive towards nucleophiles.
Statement:
Aldehydes are more reactive than ketones towards nucleophilic addition reactions.
Reasons:
1. Steric Effect
- Aldehydes have: One alkyl group + one hydrogen.
- Ketones have: Two alkyl groups.
- More alkyl groups → more steric hindrance → less reactivity.
2. Electronic Effect (+I Effect)
- Alkyl groups show +I effect.
- They donate electron density to carbonyl carbon.
- This reduces partial positive charge on carbon.
Ketones (two alkyl groups) are less electrophilic.
Order of Reactivity:
Formaldehyde > Other aldehydes > Ketones
Example:
HCHO > CH₃CHO > CH₃COCH₃
Conclusion:
Due to lower steric hindrance and higher electrophilicity, aldehydes react faster than ketones.
Statement:
Carbonyl compounds undergo reduction to alcohols or hydrocarbons depending on reagents used.
Reduction to Alcohols
Reagents:
- NaBH₄
- LiAlH₄
- Catalytic hydrogenation (H₂/Ni)
Reactions:
Aldehyde → Primary alcohol
R–CHO → R–CH₂OH
Ketone → Secondary alcohol
R–CO–R′ → R–CHOH–R′
Reduction to Hydrocarbons
1. Clemmensen Reduction:
Zn(Hg)/HCl
R–CO–R′ → R–CH₂–R′
2. Wolff–Kishner Reduction:
NH₂NH₂/KOH
R–CO–R′ → R–CH₂–R′
Key Points:
- Carbonyl group converted to CH₂ group.
- Choice of reagent depends on acidic/basic conditions.
Conclusion:
Carbonyl compounds can be selectively reduced to alcohols or completely reduced to hydrocarbons.
- Physical nature:
Ethanoic acid is a colourless liquid with a pungent vinegar-like odour and sour taste. - Glacial acetic acid:
On cooling below 289.5 K, pure ethanoic acid forms ice-like crystals and is called glacial acetic acid. - Solubility:
It is completely miscible with water, alcohol and ether, and dissolves in water with evolution of heat. - Chemical behaviour:
Ethanoic acid is a typical aliphatic carboxylic acid and shows all characteristic reactions of carboxylic acids. - Important uses:
It is used in manufacture of dyes, plastics, perfumes, acetates, esters and as table vinegar. - Acidic test:
Its aqueous solution turns blue litmus red and gives effervescence with sodium bicarbonate due to CO₂ formation. - Special tests and reactions:
It gives a wine-red colour with ferric chloride and forms ethyl acetate (fruity smell) when heated with ethanol and conc. H₂SO₄.
- Physical state: Lower aliphatic carboxylic acids are colourless liquids with pungent odour, while higher members are waxy solids due to increase in molecular mass.
- Solubility: Lower carboxylic acids are highly soluble in water due to hydrogen bonding, but solubility decreases as the alkyl chain length increases.
- Hydrogen bonding: Carboxylic acids form strong intermolecular hydrogen bonds and exist as cyclic dimers, especially in non-polar solvents.
- Boiling points: Carboxylic acids have very high boiling points compared to alcohols and hydrocarbons of similar molecular mass due to strong intermolecular hydrogen bonding.
- Melting points: The melting points of aliphatic carboxylic acids show an irregular (odd–even) pattern, where acids with even number of carbon atoms have higher melting points than adjacent odd members.
- Physical nature: Benzaldehyde is a colourless oily liquid with an almond-like odour, boiling at 452 K, and is only slightly soluble in water but soluble in organic solvents.
- General behaviour: Benzaldehyde shows most reactions of aldehydes, but its reactivity is lower than that of aliphatic aldehydes due to resonance with the benzene ring.
- Nucleophilic addition: Benzaldehyde undergoes nucleophilic addition reactions with reagents like HCN, sodium bisulphite, and Grignard reagents to form addition products.
- Condensation with ammonia derivatives: Benzaldehyde reacts with hydroxylamine, hydrazine, phenylhydrazine and 2,4-dinitrophenylhydrazine to form oxime, hydrazone, phenylhydrazone and 2,4-DNPH derivative respectively.
- Reduction: On reduction with sodium amalgam and water or with zinc and dilute HCl, benzaldehyde is converted into benzyl alcohol.
- Oxidation and tests: Benzaldehyde is oxidised to benzoic acid by strong oxidising agents and gives a silver mirror with Tollens’ reagent but does not reduce Fehling’s solution.
- Special reactions: Benzaldehyde undergoes characteristic reactions such as Tischenko reaction, Cannizzaro reaction, Perkin reaction, benzoin condensation, and Claisen–Schmidt condensation.
- Uses: Benzaldehyde is used as a flavouring agent, in the manufacture of dyes like malachite green, and as a starting material for compounds such as cinnamaldehyde, cinnamic acid and benzoyl chloride.
- Physical nature: Acetone is a colourless, volatile and highly inflammable liquid with a pleasant smell; it boils at 329 K and is completely miscible with water, alcohol and ether.
- Chemical behaviour: Acetone shows all the general chemical reactions characteristic of ketones.
- Condensation with dry HCl: In the presence of dry HCl gas, two molecules of acetone condense to form mesityl oxide, which further condenses with another acetone molecule to form phorone.
- Condensation with conc. H₂SO₄: When acetone is distilled with concentrated sulphuric acid, it undergoes condensation to form mesitylene (1,3,5-trimethylbenzene).
- Uses: Acetone is widely used as a solvent for cellulose acetate, cellulose nitrate, varnishes and lacquers, in the manufacture of chemicals like chloroform and iodoform, and for extraction of essential oils.
- Physical nature: Acetaldehyde is a colourless, volatile liquid with a pungent smell and boils at 294 K; it is completely miscible with water, alcohol and ether.
- Chemical behaviour: Acetaldehyde shows all the general chemical reactions characteristic of aldehydes.
- Polymerisation (paraldehyde): When treated with a few drops of concentrated sulphuric acid at room temperature, acetaldehyde undergoes polymerisation to form a cyclic trimer called paraldehyde.
- Polymerisation (metaldehyde): When acetaldehyde is treated with concentrated sulphuric acid at 273 K, it polymerises to form a cyclic tetramer called metaldehyde.
- Uses: Acetaldehyde is used in the manufacture of acetic acid, ethyl acetate and n-butyl alcohol, and in silvering of mirrors; paraldehyde is used in medicine as a hypnotic.
- Physical nature: Methanal (formaldehyde) is a colourless gas with a strong, pungent smell and is highly soluble in water; its 40% aqueous solution is called formalin.
- Chemical behaviour: Formaldehyde shows reactions typical of aldehydes but is more reactive and gives addition products with ammonia, bisulphite and hydroxylamine.
- Reaction with ammonia: Formaldehyde reacts with ammonia to form hexamethylenetetramine (urotropine), which is used as a urinary antiseptic.
- Polymerisation: Formaldehyde undergoes polymerisation to form paraformaldehyde, polyoxymethylene, and trioxane (metafomaldehyde) under different conditions.
- Uses: Formaldehyde (as formalin) is used as a disinfectant and germicide, for preserving biological specimens, and in the manufacture of bakelite, resins, and polymers.
- Physical state:
Lower aldehydes and ketones are colourless liquids (formaldehyde is a gas), while higher members are solids. - Odour:
Lower aldehydes have an unpleasant odour which becomes pleasant with increase in molecular mass; ketones generally have pleasant odour and some are used in perfumery. - Polarity:
Aldehydes and ketones are polar compounds due to the presence of the carbonyl (C=O) group and have high dipole moments. - Solubility:
Lower members (up to C₄) are soluble in water due to hydrogen bonding with water; solubility decreases with increase in alkyl group size. - Boiling points:
They have higher boiling points than hydrocarbons and ethers but lower than alcohols and acids, due to dipole–dipole interactions and absence of intermolecular hydrogen bonding.
- General reactivity:
Aldehydes and ketones are highly reactive due to the presence of the polar carbonyl (C=O) group. - Relative reactivity:
Aldehydes are more reactive than ketones because aldehydes have at least one hydrogen atom attached to the carbonyl carbon. - Nucleophilic addition reactions:
Both aldehydes and ketones undergo nucleophilic addition reactions as the carbonyl carbon is electron-deficient. - Addition of hydrogen cyanide:
They react with HCN to form cyanohydrins, which are useful intermediates in organic synthesis. - Addition of Grignard reagents:
Reaction with Grignard reagents (RMgX) followed by hydrolysis gives alcohols (1° from formaldehyde, 2° from other aldehydes, 3° from ketones). - Reduction reactions:
Aldehydes and ketones are reduced to alcohols using reducing agents like LiAlH₄, NaBH₄, or hydrogen in presence of catalysts. - Oxidation reactions:
Aldehydes are easily oxidised to carboxylic acids, whereas ketones resist oxidation under mild conditions.
- Reactions due to H-atom of –COOH group:
The reactions which occur due to the replacement of the acidic hydrogen atom of the carboxyl (–COOH) group are called reactions due to H-atom of the carboxyl group. - Acidic character of carboxylic acids:
The ionisation of carboxylic acids in aqueous solution to give carboxylate ions (RCOO⁻) and hydronium ions (H₃O⁺) is called the acidic character of carboxylic acids. - Reactions involving –OH part of –COOH group:
The reactions in which the –OH part of the carboxyl group is replaced by other atoms or groups such as –Cl, –OR, –NH₂ or –OCOR are called reactions involving the –OH part of the carboxyl group. - Reactions involving –COOH group as a whole:
The reactions in which the entire –COOH group participates and undergoes change are called reactions involving the carboxyl group as a whole. - Decarboxylation:
The removal of carbon dioxide (CO₂) from a carboxylic acid or its salt on heating is called decarboxylation. - Reduction of carboxylic acids:
The conversion of the –COOH group into a –CH₂OH group or into a –CH₃ group by suitable reducing agents is called reduction of carboxylic acids. - Reactions involving alkyl group (HVZ reaction):
The halogenation of the α-carbon atom of an aliphatic carboxylic acid in the presence of red phosphorus to form α-halo acids is called Hell–Volhard–Zelinsky reaction. - Reactions due to benzene ring in aromatic carboxylic acids:
The electrophilic substitution reactions occurring in the benzene ring of aromatic carboxylic acids (mainly at meta position due to –COOH group) are called reactions due to the benzene ring present in aromatic carboxylic acids.
Important Questions [27]
- Write a classification of aliphatic ketones with a suitable example.
- The product of hydrolysis of propyne in the presence of 1% HgSOA4 and 40% HA2SOA4 is ______.
- Write a reaction for the following conversion. Benzene to Benzaldehyde.
- Write preparation of propanone by using ethanoyl chloride and dimethyl cadmium.
- Ozonolysis of 2, 3 dimethyl but-2-ene, followed by decomposition by Zn dust and water gives ______.
- What is the action of Zn – Hg / conc. HCl on propanone?
- What is the action of Sodium bisulphite on propanone?
- Write Only Reactions for the Preparation of Benzophenone from Benzonitrile.
- Write chemical equation for the following:Gatterman - Koch formylation
- Write chemical equation for the following :Rosenmund reduction
- What is the action of phenyl hydrazine on propanone?
- How Will You Convert Calcium Acetate to Acetaldehyde?
- Write a Note on ‘Aldol Condensation’.
- On acid hydrolysis, propane nitrile gives.
- How is carbolic acid prepared from chlorobenzene?
- Identify ‘A' and ‘B’ in the following reaction
- Show how the following compound can be converted to benzoic acid. Phenylethene (Styrene)
- How is Methoxy Benzene Prepared from Carbolic Acid?
- What is the Action of Following Reagents on Glucose? 1. Bromine Water 2. Hydroxylamine
- The Functional Group Present in Triacylglycerol is _______.
- Explain haloform reaction with suitable example.
- How acetone is converted into propane.
- How will you prepare acetic anhydride from acetic acid?
- Identify 'A', 'B' and 'C' in following chain reaction and rewrite the chemical reactions: CHA3CHA2OH→BrA2red A′A22′PA′A→alcKCNB→EtherLiAlHA4C
- Write structure of adipic acid.
- Explain Aldol condensation of ethanal.
- Identify A and B from the following reaction: CHA3.................|....................2CHA3−C=O→Ba(OH)A2A→ΔB+HA2O
Concepts [12]
- Concepts of Aldehydes, Ketones, and Carboxylic Acids
- Classification of Aldehydes, Ketones and Carboxylic Acids
- Nomenclature of Aldehydes, Ketones and Carboxylic Acids
- Preparation of Aldehydes and Ketones
- Methods of Preparation of Carboxylic Acids
- Physical Properties
- Polarity of Carbonyl Group
- Chemical Properties of Aldehydes and Ketones
- Chemical Properties of Carboxylic Acids
- Chemical Reactions of Aldehydes and Ketones - Reactions Due to α-hydrogen
- Overview: Aldehydes, Ketones and Carboxylic Acids
- Overview of Aldehydes, Ketones and Carboxylic Acids
