Advertisements
Advertisements
Questions
How will you prepare acetic anhydride from acetic acid?
Write chemical reactions for the following conversion:
Acetic acid into acetic anhydride
Advertisements
Solution
Conversion of acetic acid into acetic anhydride:
\[\begin{array}{cc}
\phantom{...}\ce{CH3 - CO}\phantom{......................}\\
\phantom{.....}\backslash\phantom{...........}\\
\ce{\underset{(acetic acid)}{2CH3COOH} ->[P2O5][\Delta] \phantom{......}\phantom{............}O + H2O}
\phantom{................................}\\
\phantom{.....}/\phantom{...........}\\
\ce{\underset{(acetic anhydride)}{CH3 - CO}}\phantom{..................}
\end{array}\]
RELATED QUESTIONS
Benzaldehyde does NOT show positive test with ______.
Write the name of the product when ketones react with 1,2-diol in presence of dry HCl.
Explain haloform reaction with suitable example.
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Tollen’s reagent
Which of the following compounds does NOT undergo aldol condensation?
\[\begin{array}{cc} \phantom{...}\ce{R}\phantom{.................}\ce{R}\phantom{..}\ce{H}\phantom{................}\\ \phantom{...}\backslash\phantom{..................}\backslash/\phantom{..........}\phantom{.......}\\ \ce{C = O ->[H2NNH2][KOH/glycol] C + N2 + H2O}\\ \phantom{}/\phantom{............}\phantom{.......}/\phantom{}\backslash\phantom{...............}\\ \phantom{}\ce{R}\phantom{..................}\ce{R}\phantom{..}\ce{H}\phantom{...............} \end{array}\]
Reaction is called ____________.
An aldehyde or ketone reacts with hydrogen cyanide to form corresponding cyanohydrin derivatives. This reaction is an example of ____________.
Which of the following is Clemmensen reduction?
Which among the following compounds does NOT undergo aldol condensation?
Which of the following reagents is used to avoid further oxidation of aldehydes?
The formation of cyanohydrin from acetone is an example of ____________.
\[\ce{CH2 = CH2 ->[i) O3][ii) Zn/H2O] X ->[NH3] Y}\] ‘Y’ is:
Assertion: p-N, N-dimethyl amino benzaldehyde undergoes benzoin condensation
Reason: The aldehydic (−CHO) group is meta directing.
Which one of the following reaction is an example of disproportionation reaction.
Identify A, B, C and D.
\[\ce{ethanoic acid ->[SOCl2] A ->[Pd/BaSO4] B ->[NaOH] C ->[][\Delta] D}\]
How will you convert benzaldehyde into the following compound?
Benzoic acid
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Complete the following reaction.
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH2 - C - CH3 ->[HO - CH2 - CH2 - CH2 - OH][dry HCl] ?}\\
||\phantom{.........}\\
\ce{O}\phantom{.........}
\end{array}\]
How will you prepare ethyl acetate from methyl acetate?
Which of the following compound will show a positive silver mirror test?
Primary nitroalkanes are obtained in good yield by oxidising aldoximes with the help of ______.
Which one of the following substituents at para-position is most effective in stabilizing the phenoxide 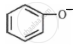 ion?
ion?

In the above reaction, product (B) is:
Which of the following is most reactive towards the addition reaction of hydrogen cyanide to form the corresponding cyanohydrin?
What is the number of different products formed when mixture of ethanal and propanal reacts with aq. NaOH after warming?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Write the structure of the products obtained from the following ketones by action of hydrazine in presence of a slightly acidic medium.
Write the structure of the products obtained from the following ketones by action of hydrazine in presence of strong base KOH.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
How will you prepare Acetic anhydride from acetic acid.
How will you prepare Acetic anhydride from acetic acid.
How will you prepare Acetic anhydride from acetic acid.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.