Advertisements
Advertisements
Question
What is the action of Zn – Hg / conc. HCl on propanone?
Advertisements
Solution
Propanone on reduction with zinc-amalgam and concentrated hydrochloric acid gives propane
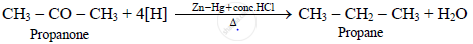
APPEARS IN
RELATED QUESTIONS
Write the structure of the product of the following reaction:
\[\ce{H3C - C ≡ C - H ->[Hg^{2+}, H2SO4]}\]
Complete the synthesis by giving missing starting material, reagent or product.

Complete the synthesis by giving missing starting material, reagent or product.
![]()
Write chemical equation for the following:
Gatterman - Koch formylation
How will you convert calcium acetate to acetaldehyde?
Complete the following reaction:
\[\ce{(C6H5CH2)2Cd + 2CH3COCI}\]
Answer the following in one sentence.
Predict the product of the following reaction:
\[\ce{CH3 - CH2 - COOCH3->[i) AIH(i-Bu)_2][ii) H3O+]}\]?
Answer the following in one sentence.
Name the product obtained by reacting toluene with carbon monoxide and hydrogen chloride in presence of anhydrous aluminium chloride.
What is the action of following reagents on ethanoic acid?
sodalime/heat
Draw structure of salicylaldehyde.
An acylchloride is hydrogenated over catalyst palladium on barium sulphate to form an aldehyde. This reaction is called as ______.
The reaction in which methyl group on benzene ring is converted to aldehydic group is called ______.
Identify 'A' and 'B' respectively in following reaction.
\[\ce{Toluene ->[Cl2][hv] A ->[H2O][\Delta] B}\]
Identify the compound A and Bin following reaction.
\[\ce{CH3Cl ->[KCN(alc)][\Delta] A ->[2H2O][HCl] B + NH4Cl}\]
A reaction in which chromyl chloride in presence of CS2 converts methyl group of toluene to aldehydic group on acid hydrolysis is called _________.
Which among the following compound on treatment with DIBAL-H yields dodecanal at 195 K?
Which of the following compounds does NOT undergo Friedel-Crafts reaction?
What is the final product obtained when benzonitrile react with phenyl magnesium bromide in equimolar proportion?
Which of the following is obtained by hydrogenation of benzoyl chloride in presence of Pd on BaSO4?
Identify product A in the following reaction:
\[\ce{Acetyl chloride + Dibenzylcadmium -> A}\]
The reaction

Which reagent is used in Etard reaction?
What is the action of hydrogen cyanide on the following?
Benzaldehyde
Write Stephen reaction for the preparation of aldehyde.
How are the following compound obtained by hydration of alkenes?
tert-butyl alcohol
Write the name of the product formed when ethanoyl chloride is treated with dimethyl cadmium.
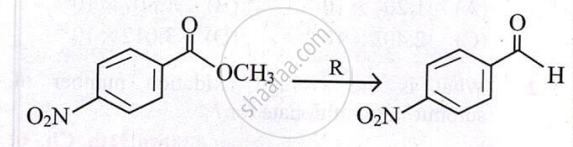
Identify the reagent R used in following transformation.