(English Medium)
Academic Year: 2024-2025
Date & Time: 21st March 2025, 11:00 am
Duration: 2h
Advertisements
- Answers to this Paper must be written on the paper provided separately.
- You will not be allowed to write during first 15 minutes.
- This time is to be spent in reading the question paper.
- The time given at the head of this Paper is the time allowed for writing the answers.
- Section A is compulsory. Attempt any four questions from Section B.
- The intended marks for questions or parts of questions are given in brackets [ ].
Which gas decolourises potassium permanganate (KMnO4) solution?
Sulphur dioxide
Ammonia
Hydrogen chloride
Carbon dioxide
Chapter:
Which formula represents a saturated hydrocarbon?
C4H8
C5H12
C4H6
C5H10
Chapter:
The metal whose oxide can be reduced by common reducing agents:
Copper
Sodium
Aluminium
Potassium
Chapter:
An organic compound has a vapour density of 22. The molecular formula of the organic compound is ______.
[Atomic weight: C = 12, H = 1]
CH4
C2H4
C2H6
C3H8
Chapter:
In the reaction given below sulphuric acid acts as a/an:
\[\ce{S + 2H2SO4 -> 3SO2 + 2H2O}\]
Non-volatile acid
Dibasic acid
Oxidising agent
Reducing agent
Chapter:
Assertion (A): The tendency of losing electrons increases down the Group.
Reason (R): The most reactive metal is placed at the top of Group 1.
Both (A) and (R) are true, and (R) is the correct explanation of (A).
Both (A) and (R) are true, and (R) is not the correct explanation of (A).
(A) is true but (R) is false.
(A) is false but (R) is true.
Chapter:
The ore that can be concentrated by using magnetic separation:
Corundum
Haematite
Calamine
Bauxite
Chapter:
The diagram given below shows the bonding in the covalent molecule AB.
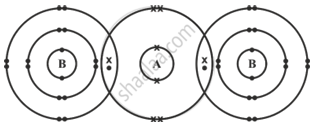
Which option represents the correct electronic configuration of atoms A and B before combining together to form the above molecule?
A - 2, 4, B - 2, 8, 6
A - 2, 4, B - 2, 8, 7
A - 2, 8, B - 2, 8, 8
A - 2, 6, B - 2, 8, 7
Chapter:
Which of the following options has all the compounds which are members of the same homologous series?
CH4, C2H6, C3H8
CH4, C2H6, C3H6
C3H4, C3H6, C3H8
C2H4, C3H6, C4H10
Chapter:
Assertion (A): In the Contact Process SO3 gas is not directly dissolved in water to obtain sulphuric acid.
Reason (R): Dense fog or misty droplets of sulphuric acid are formed which is difficult to condense.
Both (A) and (R) are true, and (R) is the correct explanation of (A).
Both (A) and (R) are true, and (R) is not the correct explanation of (A).
(A) is true but (R) is false.
(A) is false but (R) is true.
Chapter:
Given below are four ions:
Cl−, Li+, Al3+, K+
Identify the pair of ions which have the same electronic configuration.
[Atomic number: Cl = 17, Li = 3, Al = 13, K = 19]
Cl− & Li+
Al3+ & Ki+
Cl− & K+
Li+ & K+
Chapter:
Which pair of reactants can be best used to produce lead (II) sulphate?
Sulphuric acid + Lead
Sulphuric acid + Lead hydroxide
Sodium sulphate + Lead nitrate
Potassium sulphate + Lead oxide
Chapter:
Aqueous copper (II) sulphate is electrolysed using copper electrodes.
Which statement about the electrolysis is not correct?
An oxidation reaction occurs at the positive electrode.
The current is carried through the electrolyte by ions.
The positive electrode loses mass.
The number of copper (II) ions in the electrolyte decreases.
Chapter:
X, Y & Z are three metallic atoms in successive order belonging to the same group such that atomic radii of ‘X’ is the smallest. Which of the three atoms is the best reducing agent?
X
Y
Z
All three have the same reducing power.
Chapter:
40 cm3 of methane (CH4) is reacted with 60 cm3 of oxygen.
The equation for the reaction is given below:
\[\ce{CH4 + 2O2 -> CO2 + 2H2O}\]
All volumes are measured at room temperature.
What is the total volume of the gases remaining at the end of the reaction?
60 cm3
40 cm3
45 cm3
50 cm3
Chapter:
A student was instructed by the teacher to prepare and collect ammonia gas in the laboratory by using aluminium nitride. The student had set up the apparatus as shown in the diagram below. Study the given diagram and answer the following questions:
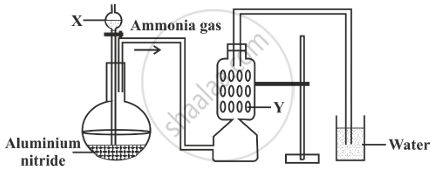
- Name the substance X added through the thistle funnel by the student.
- Write a balanced equation for the reaction occurring between Aluminium nitride and substance X.
- Identify the substance Y.
- State the function of Y.
- Why could the student not collect ammonia gas at the end of the experiment?
Chapter:
State the term for the following:
Undistilled alcohol containing a large amount of methanol.
Chapter:
State the term for the following:
A salt formed by the partial replacement of the hydroxyl group of a di-acidic or a tri-acidic base by an acid radical.
Chapter:
Give one word for the following statement:
Organic compounds having the same molecular formula but different Structural formula.
Chapter:
State the term for the following:
The tendency of an atom to attract the shared pair of electrons towards itself when combined in a compound.
Chapter:
State the term for the following:
The type of covalent bond in which electrons are shared unequally between the combining atoms.
Chapter:
______ solution forms a coloured precipitate with ammonium hydroxide which is soluble in excess of ammonium hydroxide.
Ferrous chloride
Copper nitrate
Chapter:
Advertisements
Zinc blende is converted to zinc oxide by ______.
Calcination
Roasting
Chapter:
______ conducts electricity by the movement of ions.
Molten iron
Molten sodium chloride
Chapter:
The reaction that takes place at the anode during the electrolysis of aqueous Sodium argentocyanide with silver electrodes is ______.
\[\ce{Ag -> Ag+ + e-}\]
\[\ce{Ag+ + e- -> Ag}\]
Chapter:
The salt formed when ZnO reacts with hot concentrated NaOH is ______.
sodium zincate
zinc hydroxide
Chapter:
Match the Column A with Column B:
| Column A | Column B | ||
| (a) | \[\ce{N2 + 3H2 <=> 2NH3}\] | 1. | Vanadium Pentoxide |
| (b) | \[\ce{4NH3 + 5O2 -> 4NO + 6H2O}\] | 2. | Nickel |
| (c) | \[\ce{2SO2 + O2 <=> 2SO3}\] | 3. | Iron |
| (d) | \[\ce{C2H4 + H2 -> C2H6}\] | 4. | Concentrated Sulphuric acid |
| (e) | \[\ce{CuSO4*5H2O -> CuSO4 + 5H2O}\] | 5. | Platinum |
Chapter:
Draw the structural diagram for the following organic compound:
2-methyl propene
Chapter:
Draw the structure for the following compound:
Butanal
Chapter:
Give the IUPAC name for the following organic compound:
\[\begin{array}{cc}
\ce{Cl}\phantom{....}\ce{Cl}\\
|\phantom{......}|\\
\ce{H - C - C - H}\\
|\phantom{......}|\\
\ce{Cl}\phantom{....}\ce{Cl}\\
\end{array}\]
Chapter:
Give IUPAC name for the following organic compound:
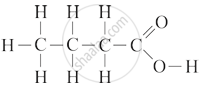
Chapter:
Give the IUPAC name for the following organic compound:
\[\begin{array}{cc}
\ce{H}\phantom{..........................}\\
|\phantom{...........................}\\
\ce{H - C - H}\phantom{....}\ce{H}\phantom{....}\ce{H}\phantom{..............}\\
|\phantom{.............}|\phantom{......}|\phantom{.......}\\
\ce{C = C - C - C - H}\\
|\phantom{......}|\phantom{......}|\phantom{......}|\phantom{......}\\
\ce{H}\phantom{.....}\ce{H}\phantom{.....}\ce{H}\phantom{.....}\ce{H}\phantom{.......}\\
\end{array}\]
Chapter:
The atomic number of two atoms ‘X’ and ‘Y’ are 14 and 8 respectively. State:
- the period to which ‘X’ belongs.
- the formula of the compound formed between ‘X’ and ‘Y’.
(Do not identify X and Y)
Chapter:
Justify the following statement:
Anode is known as the oxidizing electrode.
Chapter:
Justify the following statement:
Graphite electrodes are preferred in the electrolysis of molten lead bromide.
Chapter:
The reaction between concentrated sulphuric acid and magnesium can be represented by the equation given below:
\[\ce{Mg + 2H2SO4 -> MgSO4 + 2H2O + SO2}\]
If 60 g of magnesium is used in the reaction, calculate the following:
- The mass of sulphuric acid needed for the reaction.
- The volume of sulphur dioxide gas liberated at S.T.P.
[Atomic weight: Mg = 24, H = 1, S = 32, O = 16]
Chapter:
Give one significant observation when:
a solution of barium chloride is added to zinc sulphate solution.
Chapter:
State one observation for the following:
Lead nitrate is heated strongly in a test tube.
Chapter:
Give one significant observation when:
chlorine gas is passed over moist starch iodide paper.
Chapter:
A gas cylinder can hold 150 g of hydrogen under certain conditions of temperature and pressure. If an identical cylinder with the same capacity can hold 450 g of gas ‘G’ under the same conditions of temperature and pressure, find:
- the vapour density of the gas ‘G’.
- the molecular weight of gas ‘G’.
Chapter:
Complete and balance the following equation:
\[\ce{CH3COONa + NaOH ->[CaO][Delta]}\]
Chapter:
Complete and balance the following equation:
\[\ce{CH3COOH + Mg ->}\]
Chapter:
Name the gas produced during the following reaction:
When copper is treated with hot, concentrated nitric acid.
Chapter:
Name the gas produced during the following reaction:
When ammonia is burnt in an atmosphere of oxygen.
Chapter:
Name the gas produced during the following reaction:
When ferrous sulphide reacts with dilute hydrochloric acid.
Chapter:
Advertisements
Study the table given below. Use only the letters given in the table to answer the questions. Do not identify the elements.
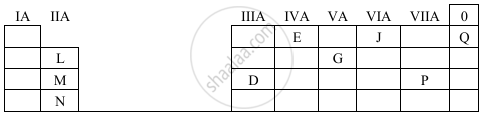
- State the valency of element ‘G’.
- Which element can exhibit catenation?
- Write the formula of the compound formed between ‘M’ and ‘P’.
Chapter:
Given below is a set of elements from the periods. Name the element with the highest ionisation potential in the following set.
Al, Cl, Mg
Chapter:
Given below is a set of elements from the periods. Name the element with the highest ionisation potential in the following set.
Ne, O, F
Chapter:
Ammonia gas is passed over heated copper (II) oxide in a combustion tube:
- Name the gas evolved.
- What will be the colour of the residue that is left in the combustion tube at the end of the reaction?
Chapter:
Give a balanced equation for the following:
Action of dilute hydrochloric acid on ammonium carbonate.
Chapter:
Give a balanced equation for the following:
Oxidation of sulphur with hot concentrated nitric acid.
Chapter:
Give a balanced equation for the following:
Reaction of concentrated sulphuric acid with carbon.
Chapter:
Rohit took two different salt solutions in test tubes C and D as shown in the figure below. He added dilute HCl to each of the two test tubes. The products formed in the test tubes C and D are silver chloride and lead chloride respectively.
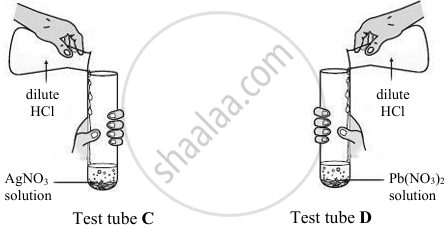
State:
- one common observation made by Rohit in both the reactions.
- the observations made by him on addition of excess of ammonium hydroxide to the products formed in:
- test tube C
- test tube D
Chapter:
Given below is a diagram showing the placement of five different oxides. With respect to the given diagram answer the following questions:
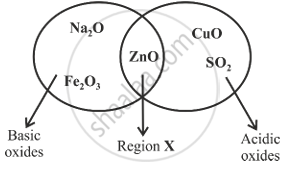
- Name the type of oxide represented in region X in the diagram.
- Identify the oxide which has been incorrectly placed in the above diagram.
- Name the oxide from the above diagram which will form an alkali when dissolved in water.
Chapter:
Given below are organic compounds labelled A to F.
Answer the questions that follow:
| A. | \[\begin{array}{cc} \phantom{..................}\ce{H}\phantom{........}\ce{O - H}\\ \phantom{..........}|\phantom{........}/\\ \ce{H - C - C}\\ \phantom{..........}|\phantom{......}\backslash\backslash\\ \phantom{...........}\ce{H}\phantom{........}\ce{O}\\ \end{array}\] |
B. | \[\begin{array}{cc} \ce{H}\phantom{.....}\ce{H}\\ |\phantom{......}|\\ \ce{H - C - C - O}\\ \phantom{.........}|\phantom{......}|\phantom{.......}\backslash\\ \phantom{..........}\ce{H}\phantom{....}\ce{H}\phantom{.......}\ce{H}\\ \end{array}\] |
| C. | \[\begin{array}{cc} \phantom{.}\ce{H}\phantom{...........}\ce{H}\\ \backslash\phantom{........}/\\ \ce{C = C}\\ /\phantom{........}\backslash\\ \phantom{.}\ce{H}\phantom{...........}\ce{H}\\ \end{array}\] |
D. | \[\begin{array}{cc} \ce{H}\phantom{....}\ce{H}\phantom{....}\ce{H}\phantom{....}\ce{H}\\ |\phantom{......}|\phantom{......}|\phantom{......}|\\ \ce{H - C - C - C - C - H}\\ |\phantom{......}|\phantom{......}|\phantom{......}|\\ \ce{H}\phantom{....}\ce{H}\phantom{....}\ce{H}\phantom{....}\ce{H}\\ \end{array}\] |
| E. | \[\begin{array}{cc} \ce{H}\phantom{....}\ce{H}\\ |\phantom{......}|\\ \ce{H - C - C - H}\\ |\phantom{......}|\\ \ce{H}\phantom{....}\ce{H}\\ \end{array}\] |
F. | \[\begin{array}{cc} \ce{H}\phantom{....}\ce{H}\phantom{....}\ce{H}\\ |\phantom{......}|\phantom{......}|\\ \ce{H - C - C - C - H}\\ |\phantom{......}|\phantom{......}|\\ \ce{H}\phantom{.....}|\phantom{.....}\ce{H}\\ \ce{H - C - H}\\ |\\ \ce{H}\\ \end{array}\] |
- Which compound forms a single product with bromine?
- Which two compounds have the same molecular formula?
- Which two compounds will react together in the presence of concentrated H2SO4 to form a product with a fruity smell?
Chapter:
An organic compound ‘X’ contains carbon, oxygen and hydrogen only. The percentage of carbon and hydrogen are 47.4% and 10.5% respectively. The relative molecular mass of ‘X’ is 76. Find the empirical formula and the molecular formula of ‘X’.
[Atomic weight: C = 12, O =16, H = 1]
Chapter:
Seema added a few pieces of copper turnings to a test tube containing concentrated acid P and she noticed that a reddish-brown gas evolved.
- Name the acid P used by Seema.
- Write a balanced chemical equation for the reaction that took place.
Chapter:
Answer the following question with reference to the concentration of bauxite ore.
Name the process used to concentrate the ore.
Chapter:
Answer the following questions with reference to the concentration of bauxite ore.
Give a balanced chemical equation for the conversion of aluminium hydroxide to pure alumina.
Chapter:
Draw the dot and cross structure of the following:
An ionic compound formed when Mg reacts with the dilute HCl.
[Atomic number: Mg = 12, Cl = 17, H = 1, N = 7]
Chapter:
Draw the dot and cross structure of the following:
A covalent compound formed when H2 reacts with Cl2.
[Atomic number: Mg = 12, Cl = 17, H = 1, N = 7]
Chapter:
Draw the dot and cross structure of the following:
The positive ion produced when ammonia gas is dissolved in water.
[Atomic number: Mg = 12, Cl = 17, H = 1, N = 7]
Chapter:
Acidulated water is electrolysed using platinum electrodes.
Answer the following questions:
- Why is dilute sulphuric acid added to water?
- Write the reaction taking place at the cathode.
- What is the observation at the anode?
Chapter:
Differentiate between the following pair of compounds using the reagent given in the bracket:
Ammonium chloride and Sodium chloride (using an alkali)
Chapter:
Differentiate between the following pair of compounds using the reagent given in the bracket:
Zinc Nitrate solution and Calcium Nitrate solution (using excess sodium hydroxide solution)
Chapter:
You are provided with some compounds in the box.

Choose the most appropriate compound which fits the descriptions (a) to (c) given below:
- A colourless gas which turns acidified K2Cr2O7 from orange to green.
- A yellow explosive oily liquid formed when excess chlorine gas reacts with ammonia gas.
- A yellow metallic oxide formed on thermal decomposition of PbCO3.
Chapter:
P, Q, R and S are the different methods of preparation of salts.
P - Simple displacement
Q - Neutralisation by titration
R - Precipitation
S - Direct combination
Choose the most appropriate method to prepare the following salts:
- PbCl2
- FeCl3
- Na2SO4
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CISCE previous year question papers ICSE Class 10 Chemistry with solutions 2024 - 2025
Previous year Question paper for CISCE ICSE Class 10 -2025 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CISCE ICSE Class 10 .
How CISCE ICSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
