Advertisements
Advertisements
Question
Given below is a diagram showing the placement of five different oxides. With respect to the given diagram answer the following questions:
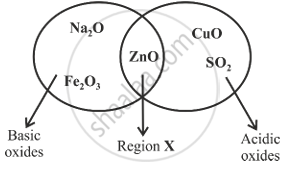
- Name the type of oxide represented in region X in the diagram.
- Identify the oxide which has been incorrectly placed in the above diagram.
- Name the oxide from the above diagram which will form an alkali when dissolved in water.
Short Answer
Advertisements
Solution
- Region X represents amphoteric oxide. It shows properties of both acidic and basic oxides.
- Copper oxide (CuO), since it is a metallic oxide and metallic oxides are basic in nature, is put in the group of acidic oxides.
- Sodium oxide will form an alkali when dissolved in water because it is water-soluble.
\[\ce{\underset{(Sodium oxide)}{Na2O}+ H2O -> \underset{Sodium hydroxide}{2NaOH}}\]
shaalaa.com
Is there an error in this question or solution?
