Advertisements
Advertisements
Question
The diagram given below shows the bonding in the covalent molecule AB.
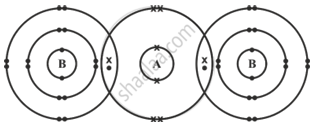
Which option represents the correct electronic configuration of atoms A and B before combining together to form the above molecule?
Options
A - 2, 4, B - 2, 8, 6
A - 2, 4, B - 2, 8, 7
A - 2, 8, B - 2, 8, 8
A - 2, 6, B - 2, 8, 7
Advertisements
Solution
A - 2, 6, B - 2, 8, 7
Explanation:
In the given diagram, atom A shares one electron with each of two atoms of B. Thus, A shares a total of two electrons, whereas B shares one electron. Element A possesses 4 unshared electrons in its outermost shell, resulting in a total of 6 electrons in that shell before any combination with other atoms. Given that A possesses only two electron shells, its electronic configuration is 2, 6. One atom of B shares one electron, resulting in a total of 7 electrons in its outermost shell, as 6 electrons remain unshared. B possesses three electron shells, resulting in an electronic configuration of 2, 8, 7.
