English Medium
Academic Year: 2025-2026
Date: March 2026
Advertisements
General Instructions:
- This question paper consists of 39 questions in 3 sections. Section A is Biology, Section B is Chemistry and Section C is Physics.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
Select the group in which all organisms have the same mode of nutrition.
Cuscuta, yeast, legumes, leeches and tapeworm
Cactus, ticks, lice, leeches and cow
Cuscuta, ticks, lice, leeches and tapeworm
Cactus, grass, lice, lion and tapeworm
Chapter:
Which of the following options indicates the products formed after breakdown of the glucose in our muscle cells when there is lack of oxygen?
Ethanol + carbon dioxide + Energy
Lactic acid + Energy
Lactic acid + carbon monoxide + Energy
Carbon dioxide + Water + Energy
Chapter:
Which of the following is a correct combination of function and part of the brain?
Posture and balance: Cerebrum
Salivation: Medulla in midbrain
Hunger: Pons in hindbrain
Blood pressure: Medulla in hindbrain
Chapter:
The blood glucose level in a patient was very high. It may be due to inadequate secretion of ______.
growth hormone from pituitary gland
oestrogen from ovary
insulin from pituitary gland
insulin from pancreas
Chapter:
In a cross between black furred rabbit (B) and white furred rabbit (b), all offspring were found to have black fur. What can be inferred about the genetic makeup of the parent rabbits?
BB × bb
Bb × Bb
Bb × bb
bb × bb
Chapter:
Which are the correct statements related to ozone?
- Ozone layer helps in increasing the UV radiations reaching earth.
- Ozone is a deadly poison.
- Ozone layer shields the earth from UV radiations.
- Ozone layer prevents UV rays which cause skin cancer.
- Ozone is formed with the help of Chloroflurocarbons.
(i), (ii), (iii)
(ii), (iii), (iv)
(iii), (iv), (v)
(i), (iv), (v)
Chapter:
Which of the following human activities has resulted in an increase of non-biodegradable substances?
Organic farming
Increase in tree plantation
Use of plastic as packaging material
Composting of kitchen waste
Chapter:
Assertion (A): Tallness of a pea plant is controlled by an enzyme.
Reason (R): The gene for that enzyme makes proteins which help the plant to be tall.
Both A and R are true, and R is the correct explanation of A.
Both A and R are true, and R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Chapter:
Assertion (A): Vulture will always have the least amount of pesticides in a food chain.
Reason (R): Vulture occupies the last trophic level and it gets only 10% of energy of the previous trophic level.
Both A and R are true, and R is the correct explanation of A.
Both A and R are true, and R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Chapter:
Unlike animals, plants do not have any excretory products as they do not eat food. Comment upon the statement with justification.
Chapter:
How many chambers are there in the heart of the following organism? How is mixing of oxygenated and deoxygenated blood prevented in its body?
Fishes
Chapter:
How many chambers are there in the heart of the following organism? How is mixing of oxygenated and deoxygenated blood prevented in its body?
Humans
Chapter:
Explain the mechanism by which the water is transported in plants?
Chapter:
About 100 acres of forest land was declared as Natural reserve park. The following organisms were predominant in the Natural reserve park:
Rabbit, frog, grass, fish, fox, water insects, zebra, peacock, snake, trees, bird, owl, insects, tiger, vulture, duck.
Create a food web comprising two separate food chains with different producers by using the above data.
Chapter:
Draw and explain how the nerve cells help in transmission of impulses?
Chapter:
Advertisements
In a genetic experiment, plants with pure round green seeds (RRyy) were crossed with plants with wrinkled yellow seeds (rrYY).
- Show the gametes formed when F1 was self-pollinated.
- A total of 144 seeds were produced which developed into saplings. Show the ratio in which these traits are independently inherited in these 144 sapling.
Chapter:
Neha consumed boiled sweet potatoes and boiled eggs for breakfast. Help her to understand some steps in the process of digestion of the food taken by her by answering the questions given below.
- Which of these food items is rich in proteins? In which part of the alimentary canal is the digestion of this component initiated? Name the enzymes, conditions required and the glands associated with the digestion here.
OR - Which of these food items contains fats? How is it digested?
- Which of these food items is rich in starch? How is its digestion initiated?
- The figure given below represents parts of the human alimentary canal. Which of these parts will have the maximum amount of digested food as soon as the process of digestion is completed?
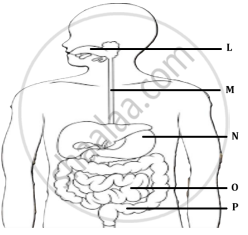
Human alimentary canal
Chapter:
Puneet wanted to grow banana plants.
- Based on your knowledge on plant reproduction should he opt for seeds or any alternate method of reproduction? Justify your answer.
- Offsprings of a banana plant usually show very little variation. What causes variation and are variations good or bad? Justify.
Chapter:
Annie was conducting research on the number of fruits produced by watermelon under different conditions. She grew 25 watermelon plants each in both glass house A and B. She introduced pollinators in glass house A only.
- What difference will she observe in the number of fruits produced in the two glass houses? Explain with reason.
- List 3 changes that will occur in a flower once it gets fertilized.
Chapter:
Which of the following equations represent redox reactions and what are the values for ‘p’ and ‘q’ in these equations?
Equation 1: \[\ce{Fe2O3_{(s)} + 2Al_{(s)} -> Al2O3_{(s)} + p Fe_{(l)} + heat}\]
Equation 2: \[\ce{2C4H10_{(g)} + 13O2_{(g)} ->[\Delta] 8CO2_{(g)} + q H2O_{(g)}}\]
Only equation 1 is a redox reaction, p = 1 and q = 3.
Both equations 1 and 2 are redox reactions, p = 2 and q = 4.
Only equation 2 is a redox reaction, p = 2 and q = 10.
Both equations 1 and 2 are redox reactions, p = 2 and q = 10.
Chapter:
Four statements about the reactions of oxides with dilute hydrochloric acid and aqueous sodium hydroxide are listed.
- Aluminium oxide reacts with both dilute hydrochloric acid and aqueous sodium hydroxide.
- Calcium oxide reacts with dilute hydrochloric acid and aqueous sodium hydroxide.
- Zinc oxide reacts with both dilute hydrochloric acid and aqueous sodium hydroxide.
- Sulphur dioxide does not react with either dilute hydrochloric acid or aqueous sodium hydroxide.
Which statements are correct?
I and II
I and III
II and IV
III and IV
Chapter:
An iron nail is added to each of the two test tubes ‘P’ and ‘Q’ containing aqueous copper (II) sulphate, and aqueous silver nitrate respectively. Which of the following observation is correct?
In test tube ‘P’ iron nail is coated with a blue coating and in test tube ‘Q’ there is no reaction.
Iron nail is coated with a brown coating in test tube ‘P’ and silver coating in test tube ‘Q’.
There is no reaction in either of the test tubes ‘P’ or ‘Q’.
There is no reaction in test tube ‘P’ but a silver coating on iron nail is seen in test tube ‘Q’.
Chapter:
Methyl orange is added to dilute hydrochloric acid and to aqueous sodium hydroxide. What is the colour of the methyl orange in each solution?
| Colour in dilute hydrochloric acid | Colour in aqueous sodium hydroxide |
| Orange | Red |
| Colour in dilute hydrochloric acid | Colour in aqueous sodium hydroxide |
| Red | Yellow |
| Colour in dilute hydrochloric acid | Colour in aqueous sodium hydroxide |
| Red | Orange |
| Colour in dilute hydrochloric acid | Colour in aqueous sodium hydroxide |
| Yellow | Red |
Chapter:
Which of the following substances when dissolved in equal volume of water, will have the highest pH value?
Sulphuric acid
Acetic acid
Magnesium hydroxide
Sodium hydroxide
Chapter:
When excess of carbon dioxide is passed through lime water, the milkiness disappears because ______.
water soluble calcium carbonate converts to water soluble calcium bicarbonate.
insoluble calcium carbonate converts to water soluble calcium bicarbonate.
water soluble calcium carbonate converts to insoluble calcium bicarbonate.
insoluble calcium carbonate converts to insoluble calcium bicarbonate.
Chapter:
In the reaction of aqueous solution of barium chloride with aqueous solution of sodium sulphate, the aqueous solution formed will be ______.
BaCl2
BaSO4
Na2SO4
NaCl
Chapter:
Assertion (A): C4H8, C4H6 and C4H10 are members of the same homologous series.
Reason (R): C4H8, C4H6, C3H4, C3H6, C2H4, C2H2 are unsaturated hydrocarbons.
Both A and R are true, and R is the correct explanation of A.
Both A and R are true, and R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Chapter:
The following activity is set-up in the science lab by the teacher. He clamped an aluminium wire on a stand and fixed a pin to the free end of the wire using wax. Then he heated the wire with a burner from the end where the wire is clamped. Students observed the pin fall off.
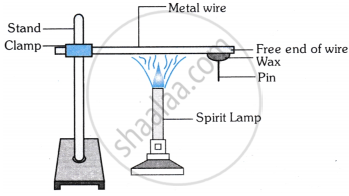
- If the teacher replaces aluminium wire by silver wire, will the students’ observation change? Justify your answer.
- Will the aluminium wire melt? Give reason for your answer.
Chapter:
An element ‘X’ is stored in kerosene, and cannot be extracted from its ore using a reducing agent. ‘X’ forms an ionic compound on reaction with chlorine.
- Can we store ‘X’ in water? Give reason to support your answer.
- Identify element ‘X’. Name the process used and write the equation for extraction of ‘X’ from its ore.
Chapter:
The domes of many building in Europe are made of copper. These domes now appear greenish in colour.
- Why do the domes appear greenish though copper is orange-red in colour?
- In your opinion, should the copper domes be replaced by iron domes to overcome the problem of change of colour of copper domes?
- Domes used to be made from thin sheets of metals. Why did the ancient architects use copper to make domes?
Chapter:
Advertisements
Amrita electrolysed distilled water using the set-up shown in figure 1. She was expecting two gases to be evolved at the anode and cathode respectively.
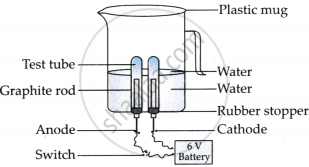
Figure 1
Suddenly, she realised that the bulb in the circuit did not glow when she used distilled water (figure 2).
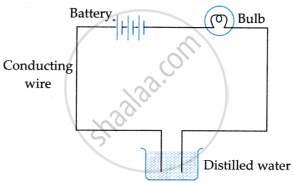
Figure 2
After this realization, she added a substance to the distilled water for electrolysis to take place.
Answer the following questions based on the information given above:
- Which gas was she expecting to be formed at the anode and which one at the cathode respectively?
- Why did the bulb not glow when Amrita passed electricity through distilled water?
- Which substance was added by Amrita to distilled water to get the expected result?
Chapter:
Sara took 2 mL of dilute NaOH solution in a test tube and added two drops of phenolphthalein solution to it. The solution turned pink in colour. She added dilute H2SO4 to the above solution drop by drop until the solution in the test tube became colourless. 40 drops of dilute H2SO4 were used for the change in colour from pink to colourless. When Sara added a drop of NaOH to the solution, the colour changed to back to pink again.
Sara now tried the activity with different volumes of NaOH and recorded her observation in the table given below:
| S. No. | Volume of dil. NaOH taken (mL) | Drops of dil. H2SO4 used |
| 1 | 2 | 20 |
| 2 | 3 | 30 |
| 3 | 4 | 40 |
Answer the following questions based on the above information:
A. If Sara used concentrated H2SO4 in place of dilute H2SO4, how many drops will be required for the change in colour to be observed?
- 40
- < 40
- > 40
Justify your answer.
B. Sara measured 20 drops of dil. H2SO4 and found its volume to be 1 mL. If Sara observed a change in colour of NaOH solution by using 3 mL of H2SO4, how many mL of NaOH did she add to the test tube initially?
OR
Sara takes 10 drops of dilute H2SO4 in the test tube and adds two drops of phenolphthalein solution to it. Then she adds NaOH dropwise. Sara observes a change in colour after adding 20 drops of NaOH. What change in colour would she observe and why?
C. Write a balanced chemical equation for the reaction taking place in the above experiment. Which of the following is true and why? The reaction is:
- neutralisation and double displacement reaction
- neutralisation and precipitation reaction
- precipitation and double displacement reaction
- neutralisation, double displacement as well as precipitation reaction.
Chapter:
A hydrocarbon with the formula CxHy undergoes complete combustion as shown in the following equation:
\[\ce{2C_xH_y + 9O2 -> 6CO2 + 6H2O}\]
- What are the values of ‘x’ and ‘y’?
- Give the chemical (IUPAC) name of the hydrocarbon.
- Draw its electron dot structure.
- Name the alcohol which on heating with conc. H2SO4 will produce the above hydrocarbon CxHy.
- Write a balanced chemical equation for the reaction of CxHy with hydrogen gas in presence of Nickel.
Chapter:
The electronic structures of atoms P and Q are shown below:
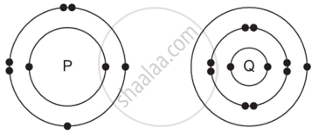
Based on the information given above, answer the following questions:
- If P and Q combine to form a compound, what type of bond is formed between them?
- Give the chemical formula of the compound formed.
- The compound so formed is dissolved in water. Is the resultant solution acidic or basic in nature? Justify your answer.
- Write the chemical equation for the reaction between ‘Q’ and ethanol.
- What will be the formula of the compound formed when ‘P’ undergoes bonding with carbon?
Chapter:
Arnav was making notes and he wrote down the following statements from his understanding of reflection from curved surfaces.
- Concave mirrors can produce both real and virtual images depending on the position of the object.
- Convex mirrors always produce real, inverted images regardless of the object’s position.
- In both concave and convex mirrors, the image location can be determined using the mirror formula `1/f = 1/v + 1/u` where f is the focal length, v is the image distance, and u is the object distance.
Choose from the following the correct option that lists the correct statements about reflection from curved surfaces.
I and II
I, II and III
II and III
I and III
Chapter:
Choose the correct option from the below which explains the reason for us to perceive the day sky as blue.
As sunlight passes through the atmosphere, shorter wavelengths, such as blue are scattered more than other colors.
The sky appears blue because all colors are scattered equally, but blue light is stronger and more visible to the human eye.
The blue color of the sky is due to longer wavelengths like red and orange scattering more than shorter wavelengths, making blue stand out more.
The atmosphere contains blue-colored particles that give the sky its blue appearance.
Chapter:
Assertion (A): A point object is placed at a distance of 26 cm from a convex mirror of focal length 26 cm. The image will not form at infinity.
Reason (R): For above given system the equation `1/f = 1/v + 1/u` gives v = ∞.
Both A and R are true, and R is the correct explanation of A.
Both A and R are true, and R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Chapter:
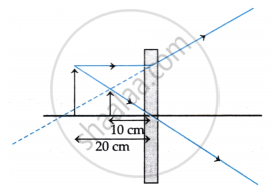
The above image shows the formation of an image with an optical instrument.
- Identify the optical instrument (shown schematically as a rectangle) in the image.
- What type of image is formed in this case?
- Based on the measurements given in the image, calculate the focal length of the instrument.
Chapter:
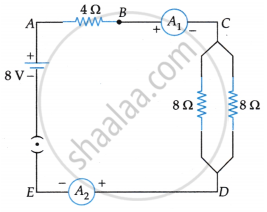
Find out the following in the electric circuit given in the figure:
- Effective resistance of two 8 ohm resistors in the combination.
- Current flowing through the 4-ohm resistor.
Chapter:
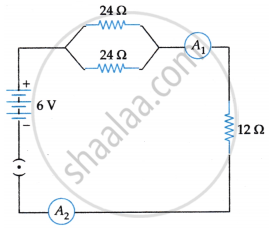
Study the circuit and find out:
- Current in 12 ohm resistor.
- Difference in the readings of ammeter A1 and A2 if any.
Chapter:
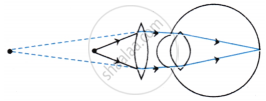
The above image shows a corrective measure for a particular defect of vision.
- Identify the defect of vision and state what kind of lens is used to correct this deficiency.
- Draw and label a ray diagram that shows the defect of vision in the above case before correction.
Chapter:
A student needs to make a 0.12 Ω resistor. She has some copper wire of 0.80 mm diameter. Resistivity of copper is 1.8 × 10–8 Ωm.
- Determine the cross-sectional area of the wire.
- Calculate the length of wire required for the 0.12 Ω resistor.
Chapter:
Magnetic field lines are shown in the given diagram. A student makes a statement that the magnetic field at X is stronger than at Y.
- Explain with reason if the student’s claim is correct.
- Also redraw the diagram and mark the direction of magnetic field lines.
Chapter:
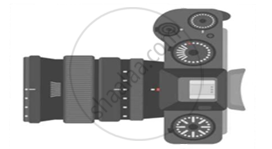

The above image is that of a Digital Single Lense Reflector (DSLR) Camera which are used to take high resolution photographs by professional photographers. The second image of the above two is a schematic diagram of how an image is formed on the sensor of the camera. Based on your understanding of the lenses, answer the following questions.
- What type of lens is used in the DSLR camera shown in the image?
- What type of image is formed on the sensor?
- A photographer is using a DSLR camera with a lens of focal length f = 50 mm to take a close-up photograph of a small object. The lens projects an image onto the camera sensor that is located 60 mm behind the lens. Calculate the object distance (i.e., the distance between the object and the lens).
OR - A photographer is using a DSLR camera to take a picture of a flower. The flower is positioned 150 mm away from the camera lens. The actual height of the flower is 80 mm, and the image height formed on the camera’s sensor is measured to be 20 mm. Calculate the focal length of the camera lens.
Chapter:
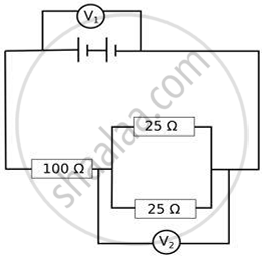
The arrangement of resistors shown in the above figure is connected to a battery.
The power dissipation in the 100 Ω resistor is 81 W. Calculate
- the current in the circuit.
- the reading in the voltmeter V2.
- the reading in the voltmeter V1.
Chapter:

An electric heater consists of three similar heating elements A, B and C, connected as shown in the figure above. Each heating element is rated as 1.2 kW, 240 V and has constant resistance. S1, S2 and S3 are respective switches.
The circuit is connected to a 240 V supply.
- Calculate the resistance of one heating element.
- Calculate the current in each resistor when only S1 and S3 are closed.
- Calculate the power dissipated across A when S1, S2 and S3 are closed.
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
CBSE previous year question papers Class 10 Science with solutions 2025 - 2026
Previous year Question paper for CBSE Class 10 Science-2026 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of CBSE Class 10.
How CBSE Class 10 Question Paper solutions Help Students ?
• Question paper solutions for Science will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
